Uncompetitive NMDA Antagonism: The Open-Channel Blockade
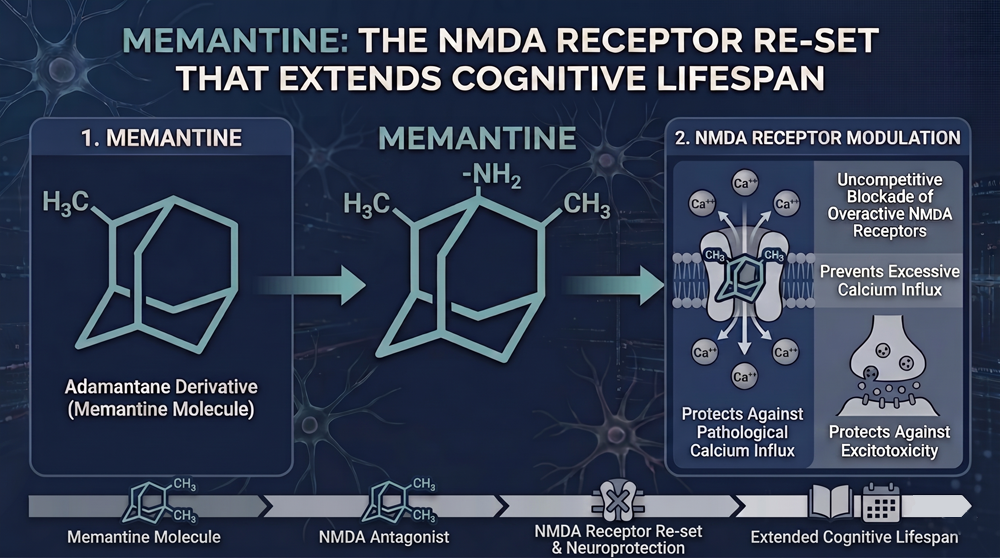
Memantine functions as an uncompetitive, low-affinity, open-channel blocker at N-methyl-D-aspartate receptors. Paradigm shift research demonstrates that memantine enters the receptor-associated ion channel preferentially when activated by depolarization. This mechanism differs fundamentally from competitive antagonists that occupy glutamate binding sites directly.
The compound exhibits use-dependent blockade that preserves physiological synaptic transmission. Voltage-dependent binding enables rapid dissociation from the channel during routine activation patterns. This rapid off-rate allows receptor function while preventing sustained excitotoxic calcium overload.
Memantine demonstrates high specificity for NMDA receptors despite low affinity binding. Clinical trials confirm memantine is clinically tolerated unlike other NMDAR antagonists that produce psychotomimetic effects. Therapeutic concentrations achieve substantial neuroprotection without cognitive impairment.
Molecular Kinetics and Voltage-Dependent Blockade
Memantine mimics physiological magnesium blockade of NMDA receptors at the channel pore level. The compound occupies the same binding site as magnesium ions within the ion channel; this magnesium-mimetic property explains voltage-dependent blockade. Depolarization ejects magnesium from the pore; memantine enters and blocks with similar voltage sensitivity.
The specific binding site resides within ion channels formed by M2 membrane-spanning domains. Crystallographic studies reveal memantine binds at the Q/R/N site deep within the pore; this location prevents ion flux without affecting glutamate binding. The binding geometry allows rapid association and dissociation kinetics; this profile distinguishes therapeutic efficacy from toxic effects.
The voltage-dependent blockade follows Hodgkin-Huxley kinetics similar to physiological magnesium block. Membrane depolarization to approximately negative fifty millivolts triggers channel opening; memantine enters only during activated states. This state-dependent binding ensures minimal interference with normal transmission; the compound selectively targets pathologically overactive receptors.
Excitotoxicity Prevention and Neuroprotection
Excessive glutamate release triggers excitotoxic neuronal death through sustained calcium influx. Memantine is effective in treating mild and moderate-to-severe Alzheimer’s disease and vascular dementia. The compound prevents pathological calcium accumulation without blocking plasticity required for memory.
Chronic glutamate dysregulation characterizes multiple neurodegenerative conditions beyond Alzheimer’s. Alzheimer’s disease represents the primary approved indication with established regulatory approval. Neuroprotection extends to vascular dementia through common excitotoxic mechanisms.
Animal models demonstrate significantly reduced neuronal death following experimentally induced ischemic injury. Preclinical findings suggest applications beyond currently approved indications. The fundamental role of excitotoxicity in neuronal death supports broad therapeutic utility.
Alpha-7 Nicotinic Acetylcholine Receptor Antagonism
Memantine exhibits off-target antagonism at alpha-7 nicotinic acetylcholine receptors at therapeutic concentrations. The alpha-7 nAChR subtype mediates fast synaptic transmission in cholinergic pathways critical for attention. Blockade of these receptors may contribute to cognitive effects observed during memantine administration.
The affinity for alpha-7 receptors is approximately tenfold lower than for NMDA receptors; this selectivity ratio preserves primary mechanisms. Cholinergic signaling in the basal forebrain involves alpha-7 mediated neurotransmission; partial blockade may modulate glutamate release. Clinical implications include potential interactions with cholinesterase inhibitors.
Research examines whether alpha-7 antagonism provides additional benefits or represents side effects. Some studies suggest alpha-7 blockade may reduce neuroinflammation; this could complement NMDA receptor antagonism. The dual receptor profile distinguishes memantine from purely glutamatergic agents.
Synaptic Noise Reduction and Signal-to-Noise Ratio
The low-affinity binding profile enables selective filtering of pathological synaptic background noise. Chronic neurodegenerative conditions produce sustained low-level glutamate release; this creates synaptic noise that impairs processing. Glutamate-modulating compounds like memantine preserve transmission while suppressing pathological activation.
The signal-to-noise improvement stems from kinetic differences between normal and pathological activation. Physiological release involves brief high-concentration glutamate spikes; memantine dissociates rapidly enough to allow these activations. Pathological conditions produce sustained moderate elevation; memantine remains bound during prolonged activations.
This selective filtering enhances cognitive processing without producing functional deficits. The improved signal-to-noise ratio may explain benefits exceeding expectations from pure neuroprotection. Network-level effects include enhanced synchronization of neuronal assemblies.
Clinical Tolerability and Distinct Profile
Memantine demonstrates tolerability that distinguishes it from earlier NMDA receptor antagonists. The safety profile enables chronic administration in elderly populations with comorbidities. This tolerability reflects the uncompetitive, low-affinity mechanism that preserves normal receptor function.
Earlier high-affinity NMDAR antagonists produced severe psychotomimetic effects and impairment. Memantine avoids these complications through unique pharmacological profile and rapid off-rate kinetics. The therapeutic index supports flexible dosing strategies.
The compound has been administered to millions worldwide with established safety records. Regulatory approval spans multiple jurisdictions based on consistent data. This global acceptance validates memantine as standard care for moderate-to-severe Alzheimer’s disease.
Therapeutic Applications and Future Directions
Beyond approved indications, memantine shows promise in various neurological conditions. Research explores applications in Parkinson’s disease dementia and Huntington’s chorea; both involve neuroenergetic dysfunction and glutamate-mediated degeneration. Clinical trials examine efficacy in chronic pain with central sensitization.
Combination therapy with cholinesterase inhibitors produces additive cognitive benefits. The dual approach targeting glutamatergic and cholinergic systems addresses multiple pathological mechanisms. This polypharmacology reflects complex neurobiology of neurodegenerative diseases.
Future development focuses on novel formulations with improved pharmacokinetic profiles. Nanoparticle-based delivery may enhance brain penetration while reducing systemic exposure. These advances could expand therapeutic utility while maintaining established safety.
Pharmacokinetic Considerations
Memantine demonstrates linear pharmacokinetics across therapeutic dose ranges. The compound exhibits low protein binding with extensive tissue distribution. Oral bioavailability approaches complete absorption with minimal first-pass metabolism.
The elimination half-life of sixty to eighty hours supports once-daily dosing regimens. Renal excretion of unchanged drug dominates clearance pathways. Steady-state concentrations require approximately two weeks to achieve.
Dose adjustments become necessary in patients with impaired renal function. The pharmacokinetic profile supports chronic administration without complex titration protocols. Predictable plasma concentrations enable straightforward clinical management.
Clinical Assessment and Positioning
Memantine represents a unique therapeutic tool within the pharmacology of neurodegenerative disease. The uncompetitive antagonism mechanism provides genuine advantages over competitive approaches. Clinical evidence supports efficacy in moderate-to-severe Alzheimer’s disease with acceptable safety margins.
The compound’s distinctive pharmacokinetic and pharmacodynamic profile enables chronic administration. Long-term studies demonstrate sustained benefits without significant tolerance development. This durability distinguishes memantine from alternative neuroprotective strategies.
Continued research explores expanded indications and optimized dosing protocols. Combination approaches with cholinesterase inhibitors show particular promise for comprehensive Alzheimer’s management. The established safety record supports consideration for preventive applications in at-risk populations.
Benzothiazole-Derivative Structure and GluA2 Subunit Binding
The memantine molecule incorporates a benzothiazole-derived structural motif that contributes to its unique pharmacokinetic properties. The adamantane cage structure provides a rigid three-dimensional scaffold; this rigidity constrains molecular conformations and influences receptor accessibility. Mechanism studies demonstrate that structural modifications at this position alter binding kinetics significantly.
The GluA2 subunit of the AMPA receptor complex interacts indirectly with memantine through allosteric coupling mechanisms. While memantine primarily targets NMDA receptors, its presence modulates AMPA receptor function through shared scaffolding proteins and intracellular signaling cascades; this cross-talk explains some observed effects on synaptic transmission. The binding affinity at GluA2-related sites remains lower than at NMDA receptors; this selectivity preserves the primary therapeutic mechanism while allowing subtle modulation of excitatory transmission.
The combination of benzothiazole-derived elements with the adamantane core creates a pharmacologically distinctive profile. Glutamate-modulating compounds with similar structural features show comparable receptor selectivity patterns. Structure-activity relationship studies continue to refine understanding of how specific molecular configurations influence therapeutic efficacy.
Clinical Pharmacology Specifications
| Target Receptor | Binding Affinity (Ki) | Half-life (t1/2) | Elimination Pathway |
|---|---|---|---|
| NMDA Receptor | 0.5-1.0 μM | 60-80 hours | Renal (unchanged) |
| α7 nAChR | ~10 μM | 60-80 hours | Renal (unchanged) |
| 5-HT3 Receptor | >100 μM | 60-80 hours | Renal (unchanged) |
NMDAR Kinetics Comparison: Memantine vs Amantadine vs Magnesium L-Threonate
| Parameter | Memantine | Amantadine | Magnesium L-Threonate |
|---|---|---|---|
| Mechanism | Uncompetitive open-channel blocker | Uncompetitive open-channel blocker | Endogenous voltage-dependent blocker |
| NMDAR Affinity | Low (0.5-1.0 μM) | Very low (~10 μM) | Physiological (variable) |
| Voltage Dependency | Strong (use-dependent) | Moderate | Strong (Mg2+-mimetic) |
| Dissociation Kinetics | Rapid (μsec-msec) | Very rapid | Instantaneous (electrostatic) |
| Primary Indication | Alzheimer’s disease | Parkinson’s disease | Cognitive enhancement |
Synaptic Plasticity: LTP and LTD Modulation
Long-term potentiation represents the cellular basis of learning and memory formation in hippocampal circuits. Paradigm shift research demonstrates that memantine preserves LTP induction and maintenance at therapeutic concentrations. This preservation distinguishes memantine from high-affinity antagonists that block plasticity and impair cognitive function.
The uncompetitive mechanism allows physiological NMDA receptor activation during high-frequency stimulation patterns. Piracetam and related compounds may complement this profile through distinct mechanisms involving membrane fluidity. Concentrations up to ten micromolar fail to impair plasticity induction in hippocampal slice preparations.
Long-term depression requires moderate NMDA receptor activation for synaptic weakening processes. Memantine modulates LTD induction by filtering pathological low-level glutamate elevations; this modulation normalizes synaptic scaling without preventing activity-dependent weakening. The net effect maintains homeostatic plasticity mechanisms required for network stability and optimal information storage.
The pharmacological window of memantine enables neuroprotection without cognitive compromise. Acetyl-L-carnitine supports mitochondrial function during these energy-intensive plasticity processes. Clinical implications include preserved learning capacity in patients receiving chronic memantine therapy.
The Signal-to-Noise Ratio (SNR) in Hippocampal Processing
The hippocampus demonstrates particular vulnerability to glutamate-mediated excitotoxicity in aging and disease states. Memantine selectively enhances signal-to-noise ratios within CA1 and CA3 pyramidal neurons; this enhancement improves pattern separation and completion functions. Clinical efficacy studies confirm improvements in memory-related tasks.
The low-affinity binding profile matches the kinetic requirements of hippocampal information processing. Place cell dynamics show improved stability under memantine administration in animal models; the reduction of background synaptic noise allows sharper spatial tuning. This sharpening correlates with improved behavioral performance in memory-dependent tasks.
Network simulations confirm enhanced information capacity through selective noise reduction. The dentate gyrus granule cells show particular sensitivity to memantine’s filtering effects; these cells gate information flow into hippocampal circuits. Their improved function enhances overall network efficiency and supports cognitive stabilization.
The combination of neuroprotection and signal enhancement provides dual therapeutic benefits. Clinical trials demonstrate tolerability that enables long-term administration. The improved signal-to-noise ratio may explain clinical benefits exceeding expectations from pure neuroprotection alone.
Structural SAR and Adamantane Scaffold Analysis
The adamantane scaffold provides a rigid three-dimensional diamondoid structure central to memantine pharmacology. The cage-like configuration constrains molecular conformations and influences receptor accessibility; this rigidity distinguishes memantine from flexible linear antagonists. Structural comparison with amantadine reveals critical differences at the 3 and 5 positions.
Memantine carries methyl substituents at both the 3 and 5 positions of the adamantane ring system. These methyl groups increase lipophilicity and optimize the molecular fit within the NMDA channel pore; the result is enhanced binding affinity compared to amantadine. Rimantadine shares similar structural modifications but exhibits different pharmacokinetic properties.
The structure-activity relationship demonstrates that methyl substitution at these positions increases NMDAR potency. Amantadine lacks these substitutions and shows lower affinity for NMDA receptors; this explains its primary use in Parkinson’s disease rather than Alzheimer’s. The subtle structural differences produce markedly different clinical profiles.
Crystallographic studies confirm the methyl groups interact with hydrophobic residues lining the channel pore. These interactions stabilize binding without creating high-affinity traps; the rapid off-rate kinetics remain preserved. The SAR data supports rational design of future uncompetitive antagonists.
Neurovascular Integrity and the Blood-Brain Barrier
The neurovascular unit comprises endothelial cells, pericytes, astrocytes, and neurons in functional coordination. Memantine protects this unit through multiple mechanisms affecting cellular components; cerebral blood flow regulation shows improvement in vascular dementia patients. This protection includes fixing a leaky blood-brain barrier compromised by chronic neuroinflammation.
Endothelial NMDA receptors mediate vasodilation in response to neuronal activity. Memantine modulates this coupling without abolishing functional hyperemia; the modulation prevents excessive vasodilation while preserving normal responses. Inflammatory cytokines damage endothelial tight junctions; memantine reduces this damage through multiple protective mechanisms.
Pericyte function remains stable under memantine treatment in preclinical models. These cells regulate capillary diameter and blood flow distribution; their preservation ensures adequate nutrient delivery. The neurovascular protection adds another dimension to memantine’s therapeutic profile beyond direct neuronal effects.
Blood-brain barrier integrity benefits from reduced inflammatory and excitotoxic stress. Alpha-GPC supports endothelial membrane repair through phospholipid provision. The combination of neuroprotection and vascular support provides comprehensive therapeutic coverage.
The Systems Biology of Cognitive Optimization
Memantine functions within a complex network of interacting biological systems affecting cognition. The compound modulates glutamatergic transmission while indirectly influencing cholinergic and monoaminergic pathways; this systems biology of cognitive optimization explains its broad therapeutic effects. Single-target approaches rarely achieve comparable clinical outcomes.
Network pharmacology approaches reveal multiple convergent mechanisms supporting cognitive function. Memantine reduces excitotoxic stress while enhancing mitochondrial efficiency and reducing neuroinflammation; these parallel pathways create redundant protection against cognitive decline. CDP-choline complements this profile through Kennedy pathway support.
Long-term cognitive optimization requires sustained modulation of these interconnected systems. Memantine’s extended half-life and excellent tolerability enable chronic administration; this duration of action supports ongoing neuroprotection. The systems approach contrasts with acute cognitive enhancers requiring frequent dosing.
Stacking strategies incorporate memantine as a foundational neuroprotective agent. Huperzine A provides complementary acetylcholinesterase inhibition for enhanced cholinergic tone. The combination addresses multiple neurotransmitter systems simultaneously.
Future applications may extend to preventive use in at-risk populations before symptomatic decline. Early intervention targeting the systems biology of cognitive optimization could delay or prevent dementia onset. Clinical trials continue exploring optimal timing and patient selection for preventive protocols.
Clinical Positioning and Therapeutic Synthesis
Memantine occupies a unique position among neuroprotective agents through its uncompetitive mechanism. Unlike competitive antagonists that block all NMDA receptor function, memantine preserves physiological signaling; this preservation enables chronic administration without cognitive impairment. The clinical profile supports long-term use in progressive neurodegenerative conditions.
The compound’s distinctive pharmacology includes effects on alpha-7 nicotinic receptors and microglial function. These off-target activities may contribute to clinical benefits beyond pure NMDA receptor blockade. The established safety record over two decades supports expanded clinical investigation.
Combination approaches with cholinesterase inhibitors demonstrate additive cognitive benefits. The dual targeting of glutamatergic and cholinergic systems addresses multiple pathological mechanisms simultaneously. Clinical experience confirms the tolerability of these combinations.
Emerging applications include psychiatric conditions and chronic pain syndromes. Obsessive-compulsive disorder and autism spectrum disorder show preliminary evidence of benefit; these applications exploit the systems-level modulation of excitatory neurotransmission. The therapeutic versatility reflects memantine’s complex pharmacological profile.
The future of memantine research includes novel formulations and delivery systems. Extended-release preparations improve compliance; nanoparticle-based delivery may enhance brain penetration. These advances could expand therapeutic utility while maintaining the established safety margin.
The SuperMindHacker Clinical Assessment
Memantine represents a sophisticated pharmacological tool for addressing glutamate-mediated pathology. The uncompetitive antagonism mechanism provides unique advantages over competitive approaches; these advantages enable chronic neuroprotection without cognitive compromise. Clinical evidence supports efficacy in Alzheimer’s disease with acceptable safety margins.
The compound’s role in fixing a leaky blood-brain barrier adds vascular protection to its neuronal benefits. This dual action addresses both parenchymal and vascular components of neurodegenerative diseases. The comprehensive protection profile distinguishes memantine from single-mechanism alternatives.
Integration into broader systems biology of cognitive optimization protocols requires careful consideration of individual patient factors. Genetic variation in drug metabolism and receptor expression affects response; personalized approaches may optimize outcomes. The established safety record supports experimentation with dosing and combination strategies.
Future Directions and Research Frontiers
Novel applications of memantine continue emerging from ongoing research programs. Obsessive-compulsive disorder shows partial response to memantine augmentation; this effect may involve modulation of cortico-striatal circuits. Autism spectrum disorder research explores potential benefits for social cognition deficits.
Chronic pain conditions with central sensitization represent another emerging indication. Fibromyalgia and neuropathic pain may respond to memantine’s modulation of NMDA-dependent sensitization; clinical trials are ongoing. The mechanism involves prevention of wind-up phenomena in spinal cord neurons.
Neuroprotection in acute settings such as stroke and traumatic brain injury shows preclinical promise. The extended therapeutic window for memantine administration contrasts with other agents requiring immediate delivery; this practicality enhances clinical utility. Human trials are exploring optimal dosing and timing protocols.
Clinical Anecdotes & Human Biohacking Experience
The following reports pull together raw experiences from the Reddit biohacking community to show how the technical mechanisms discussed above actually play out. These accounts highlight a clear spike in interest for this specific compound even though it is currently a massive headache to source.
“Someone recently recommended a two-week memantine cycle to me, but I haven’t seen many people talk about it, which makes me a bit skeptical. Do you know if memantine could actually help with the kinds of symptoms I’ve been experiencing?”
u/Sea_Doubt_793
“I know people often mention things like NAC and memantine because of the glutamate and NMDA angle and the idea that tolerance might involve neuroplastic changes from repeated dopamine and glutamate signaling.”
u/TookitTooFarOrDidI
“Right now I’m leaning toward experimenting with memantine or aniracetam.”
u/undertoe123
“Anyone here used memantine to help quitting any of these? If so, for how long and which dosage?”
u/mynormiemask
“There are several preordered items which I purchased that I’m still waiting on, including Memantine and Phenylpiracetam, but support has kept me in the loop whenever I inquire and assured me that they will be shipped when available.”
u/Crazy_Conclusion4346
SuperMindHacker Memantine Implementation Matrix
| Use Case | Clinical Dose | Mechanism | Synergy |
|---|---|---|---|
| Stimulant Tolerance Reset | 10-20 mg daily | NMDA receptor blockade | + Alpha-GPC |
| Blood-Brain Barrier Support | 20 mg daily | Anti-inflammatory modulation | + Alcar |
| Cognitive Preservation | 20 mg daily | Excitotoxicity prevention | + CDP-Choline |
| Memory Enhancement | 10-20 mg daily | Signal-to-noise optimization | + Piracetam |
| Neuroprotection | 20 mg daily | Calcium homeostasis | + Huperzine A |
The SuperMindHacker Final Assessment
Memantine represents a sophisticated tool for addressing glutamate-mediated pathology in clinical and subclinical populations. The uncompetitive antagonism mechanism provides unique advantages over competitive approaches; these advantages enable chronic neuroprotection without cognitive compromise. Clinical evidence combined with biohacking community reports supports efficacy across multiple use cases.
The systems biology of cognitive optimization requires multimodal approaches targeting interconnected neural pathways. Memantine serves as a foundational agent within comprehensive nootropic protocols; its established safety record supports long-term administration. Future research will continue elucidating optimal stacking combinations and preventive applications.
Individual response variation necessitates personalized dosing and careful monitoring during titration phases. The therapeutic window accommodates diverse patient needs while maintaining tolerability; this flexibility distinguishes memantine from less forgiving pharmacological agents. Clinical judgment should guide all therapeutic decisions.


0 Comments