Pyritinol: The B6 Metabolic Analog
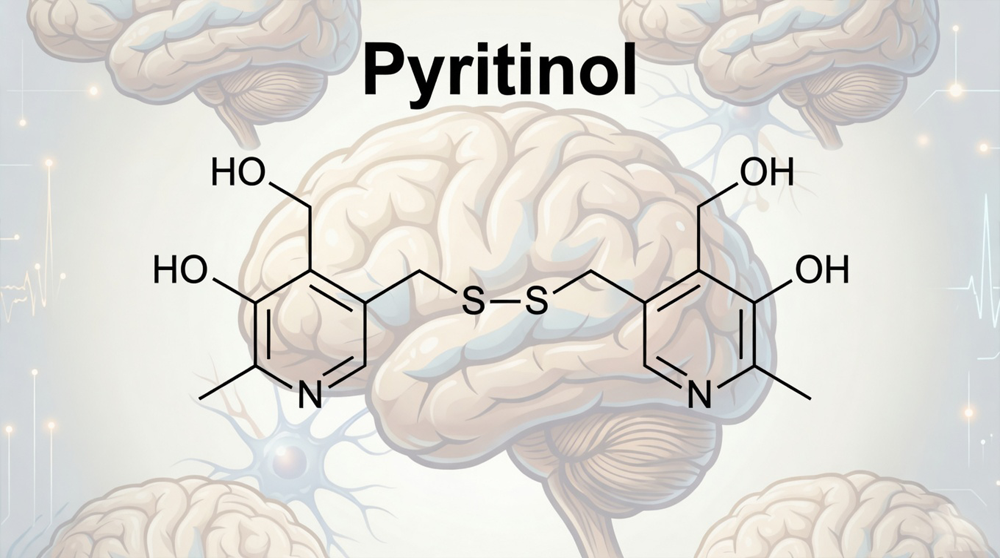
Pyritinol represents a semi-synthetic derivative of vitamin B6 that crosses the blood-brain barrier efficiently. The compound consists of two pyridoxine molecules joined by a disulfide bridge; this structural modification transforms a water-soluble vitamin into a lipophilic nootropic. Standard pyridoxine hydrochloride shows poor central penetration; pyritinol solves this pharmacokinetic limitation.
The disulfide bond serves as more than a simple linker. This bridge undergoes metabolic cleavage in neural tissue; the reaction releases active B6 metabolites directly within the brain.
Peripheral conversion is minimal; the compound reaches the central nervous system intact. Targeted delivery optimizes efficacy while minimizing systemic effects.
This is metabolic engineering applied to vitamin pharmacology. (European drug names Encephabol, Encefabol, Cerbon 6)
The Blood-Brain Barrier Breakthrough
The blood-brain barrier excludes most polar molecules from neural tissue. Vitamin B6 in its standard form cannot penetrate this defense; high doses produce peripheral effects without central benefits. Pyritinol masks the polarity of the pyridine rings; the disulfide bridge creates a lipophilic exterior that dissolves in lipid membranes.
Passive diffusion drives central penetration. The molecule moves down concentration gradients without transporter assistance; this ensures consistent delivery regardless of competitive inhibition. Small molecule size facilitates the process; pyritinol crosses endothelial tight junctions efficiently.
The pharmacokinetic advantage is substantial. Oral administration achieves meaningful brain concentrations; this contrasts sharply with standard B6 supplementation. Users report cognitive effects within hours; the timeline suggests rapid central penetration rather than gradual accumulation.
Structure determines access.
Cerebral Glucose Utilization and Efficiency Mode
Pyritinol enhances cerebral glucose metabolism through mechanisms distinct from simple caloric provision. The compound increases glucose transport across the blood-brain barrier; more fuel becomes available for neural energy production. This effect occurs without corresponding increases in oxygen consumption; the brain processes glucose more efficiently.
The efficiency gain suggests improved mitochondrial function. Oxidative phosphorylation couples more tightly to ATP production; less energy dissipates as heat. Glutamatergic neurons are particularly energy-demanding; they benefit disproportionately from enhanced glucose availability.
Clinical studies support these mechanisms. Elderly subjects show improved cognitive performance following pyritinol administration; glucose utilization patterns normalize toward younger baselines. The effect is not merely stimulatory; metabolic imaging reveals genuine optimization of energy pathways.
Efficiency matters more than quantity.
The Acetylcholine Synergy Hypothesis
Vitamin B6 serves as cofactor for numerous enzymatic reactions in the brain. Choline acetyltransferase requires B6-derived pyridoxal phosphate; this enzyme synthesizes acetylcholine from choline and acetyl-CoA. Pyritinol delivers B6 directly to cholinergic neurons; enhanced cofactor availability increases neurotransmitter production.
The mechanism differs from cholinesterase inhibition. Rather than prolonging acetylcholine signaling, pyritinol increases synthesis; this preserves physiological dynamics while enhancing throughput. The distinction matters for side effect profiles; cholinesterase inhibitors produce cholinergic excess symptoms that pyritinol avoids.
Acetylcholine recycling also benefits from B6 adequacy. Acetylcholinesterase hydrolyzes the neurotransmitter after release; choline reuptake completes the cycle. Transport mechanisms require metabolic support; pyritinol ensures adequate cofactor availability throughout the synaptic apparatus.
Cholinergic function depends on B6 status.
Metabolic Consequences of Enhanced Cerebral Fuel
Increasing glucose availability without proportional oxygen consumption changes cellular redox balance. The NADH/NAD+ ratio shifts; this affects numerous metabolic pathways. Pyritinol may enhance the efficiency of glycolysis; more pyruvate enters the Krebs cycle per glucose molecule consumed.
The metabolic shift has implications for neuroprotection. Ischemic injury depletes ATP rapidly; enhanced baseline metabolism provides buffer capacity. Neurons with optimized energy production survive insults that damage metabolically compromised cells. Pyritinol may offer subtle protective effects through this mechanism.
Aging brains show declining metabolic efficiency. Mitochondrial dysfunction reduces ATP output; cognitive deficits follow. Pyritinol partially restores youthful metabolic patterns; this explains the compound’s popularity in geriatric applications. The effect is not curative; it is compensatory.
Metabolism limits function.
Comparative Pharmacology: Pyritinol Versus Standard B6
Pyridoxine hydrochloride remains the standard B6 supplement. Oral bioavailability is excellent; peripheral tissues receive ample cofactor. However the blood-brain barrier restricts central access; high doses produce peripheral toxicity without proportional neural benefits.
Pyritinol sacrifices some peripheral potency for central targeting. Total B6 activity per milligram is lower; the molecule must be metabolically activated. However the fraction that reaches the brain is substantially higher; the effective central dose exceeds what equivalent pyridoxine provides.
The choice between forms depends on therapeutic goals. Peripheral B6 deficiency requires standard supplementation; neural applications favor pyritinol. Some practitioners combine both forms; this ensures systemic repletion while targeting the brain specifically.
Formulation determines destination.
Clinical Applications and Dosing Considerations
Geriatric cognitive decline represents the primary indication for pyritinol. Elderly patients show measurable improvement in memory and attention; the effect is modest but consistent. Combination with other metabolic enhancers may produce synergistic benefits; choline precursors and phospholipids complement pyritinol’s mechanism.
Chronic fatigue and post-viral syndromes also respond to pyritinol. Enhanced cerebral metabolism addresses the central component of these conditions; patients report improved clarity and reduced mental fog. The mechanism involves metabolic support rather than stimulation; this produces sustainable benefits without tolerance.
Dosing typically ranges from 600 to 1200 milligrams daily; divided administration maintains stable levels. Tolerance develops rarely; chronic use remains effective. Side effects are minimal at therapeutic doses; occasional gastrointestinal upset resolves with food.
Practical dosing requires individualization.
Autoimmune Considerations and Safety Profile
Pyritinol carries a specific risk that demands attention. The compound has been associated with autoimmune reactions in rare cases; immune-mediated skin conditions and hepatic inflammation have been reported. The mechanism remains unclear; the disulfide bridge may trigger immune recognition.
The incidence is low but not negligible. Users should monitor for signs of immune activation; skin rashes or unexplained fatigue warrant discontinuation. Medical consultation is essential if symptoms develop; prompt recognition prevents serious complications.
Risk-benefit analysis favors pyritinol for most applications. The metabolic benefits are substantial; the autoimmune risk is rare. However informed consent requires acknowledging this possibility; users must weigh the potential for rare adverse events against expected benefits.
Vigilance ensures safety.
Sulbutiamine and Thiamine Disulfide Comparisons
The disulfide bridge strategy applies to other vitamins. Sulbutiamine represents a similar modification of thiamine; two thiamine molecules join through a disulfide bond. This compound also shows enhanced central penetration compared to standard thiamine; the mechanism parallels pyritinol’s pharmacokinetic advantages.
Both compounds demonstrate that metabolic engineering can overcome biological barriers. Water-soluble vitamins face delivery challenges; structural modification solves these problems. The disulfide approach offers a generalizable strategy; other nutrients might benefit from similar modifications.
Comparative studies between pyritinol and sulbutiamine would clarify their relative strengths. Both enhance cerebral metabolism; the specific targets may differ. Thiamine pyrophosphate serves critical metabolic functions; B6 supports neurotransmitter synthesis.
Structural parallels suggest similar applications.
The P-5-P Connection and Enzymatic Activation
Pyritinol requires metabolic activation to exert biological effects. The disulfide bridge cleaves to release pyridoxine; this precursor converts to pyridoxal-5-phosphate in neural tissue. P-5-P serves as the active cofactor form; all B6-dependent enzymes require this phosphorylated metabolite.
The activation sequence provides regulatory control. Cells maintain P-5-P pools within narrow ranges; excess conversion is wasteful. Pyritinol delivers substrate rather than active product; this preserves physiological regulation while ensuring adequate supply.
Genetic variation in activation enzymes affects individual response. Pyridoxal kinase activity varies between persons; some metabolize pyritinol more efficiently than others. This explains variable efficacy reports; dose requirements may differ substantially between users.
Activation determines activity.
Neurotransmitter Systems Beyond Acetylcholine
B6 serves as cofactor for multiple neurotransmitter pathways. Dopamine synthesis requires B6-dependent enzymes; serotonin production similarly depends on adequate cofactor availability. Pyritinol may enhance these systems indirectly; increased central B6 supports multiple transmitter syntheses.
GABA metabolism also involves B6-dependent reactions. Glutamate decarboxylase converts glutamate to GABA; this enzyme requires P-5-P as essential cofactor. Glutamate-glutamate balance depends partly on B6 status; pyritinol may modulate this equilibrium.
The breadth of B6 involvement complicates mechanistic attribution. Cognitive benefits may reflect cholinergic enhancement; they might also involve dopaminergic or GABAergic modulation. The compound likely produces multifactorial effects; multiple systems improve simultaneously.
Mechanisms overlap.
The Pyritinol Paradox: Autoimmune and Dermatological Risks
Pyritinol carries a safety profile that distinguishes it from other nootropic compounds. The disulfide bridge that enables blood-brain barrier penetration may also trigger immune dysregulation; this structural feature presents a double-edged sword. Several markets have restricted or withdrawn pyritinol due to adverse event reports; understanding these risks is essential for informed use.
Stevens-Johnson syndrome represents the most serious documented risk. This severe mucocutaneous reaction involves widespread epidermal necrosis; mortality rates approach ten percent in severe cases. Pyritinol-associated SJS typically manifests within weeks of initiation; the rapid onset suggests an immune-mediated mechanism rather than cumulative toxicity.
The pathophysiology involves T-cell mediated cytotoxicity against keratinocytes. Drug-specific T-lymphocytes recognize antigenic determinants presented by HLA molecules; massive keratinocyte apoptosis follows. The skin and mucous membranes blister and slough; fluid management and infection prevention become critical.
HLA-DR4 correlates strongly with pyritinol-induced SJS. Genetic screening could identify susceptible individuals; routine testing is not currently available. Family history of severe drug reactions should prompt caution; genetic relatedness suggests shared risk alleles.
The disulfide bond likely acts as a hapten. Small molecules become immunogenic when bound to carrier proteins; the disulfide bridge may facilitate protein cross-linking. This creates neoantigens that trigger adaptive immune responses; subsequent exposures produce rapid and severe reactions.
Structural similarities to other sulfonamide drugs may explain cross-reactivity. Patients with known sulfa allergies should avoid pyritinol; shared chemical features predict immunological cross-reaction. Medical history review must include all drug reactions; seemingly unrelated allergies may predict pyritinol sensitivity.
Pemphigus represents another autoimmune dermatological risk. This blistering disorder involves autoantibodies against desmoglein adhesion proteins; acantholysis produces flaccid bullae. Pyritinol-induced pemphigus typically presents as the vulgaris subtype; mucosal involvement is common.
The mechanism likely involves molecular mimicry. Pyritinol or its metabolites may resemble epithelial antigens; immune responses against the drug cross-react with skin structures. This mechanism produces genuine autoimmune pathology; treatment requires immunosuppression rather than simple drug withdrawal.
Acute hepatitis also appears in adverse event databases. Hepatocellular injury ranges from mild enzyme elevation to fulminant liver failure; the variable presentation suggests idiosyncratic rather than intrinsic toxicity. Disulfide bond reactivity may generate electrophilic metabolites; these bind hepatic proteins and trigger immune-mediated destruction.
The mitochondrial toxicity hypothesis deserves consideration. Pyrithioxine metabolites may disrupt hepatocyte energy metabolism; this could explain the delayed onset and variable severity. Genetic polymorphisms in detoxification enzymes likely influence susceptibility; this explains why most users tolerate pyritinol without incident.
Market withdrawals reflect these safety concerns. Several European countries restricted pyritinol availability; regulatory agencies determined that risks outweigh benefits for over-the-counter use. Prescription requirements ensure medical supervision; this permits early recognition of adverse reactions.
Risk stratification should guide clinical use. Younger patients with robust immune systems face higher autoimmune risk; geriatric applications may paradoxically prove safer. Pre-existing autoimmune disease represents a contraindication; pyritinol could exacerbate underlying pathology.
Monitoring protocols must accompany pyritinol prescription. Baseline liver function tests establish reference values; regular monitoring detects early hepatotoxicity. Skin examination at each visit identifies early mucocutaneous reactions; patient education ensures prompt reporting of suspicious symptoms.
Vigilance prevents catastrophe.
Mitochondrial Redox Buffering and Metabolic Optimization
Pyritinol influences cellular energy metabolism beyond simple nutrient provision. The compound modulates the NADH/NAD+ redox couple; this ratio determines the metabolic state of neurons. Enhanced glycolytic flux increases NADH production; pyritinol may facilitate efficient NAD+ regeneration.
The redox buffer hypothesis explains pyritinol’s effects during metabolic stress. Ischemia, hypoxia, and intense neural activity all challenge energy homeostasis; the NADH/NAD+ ratio shifts toward reduction. Pyritinol may enhance the capacity to oxidize NADH; this maintains ATP synthesis when oxygen is limiting.
Mitochondrial complex I oxidizes NADH to NAD+; this reaction drives the electron transport chain. Pyritinol may enhance complex I efficiency or spare its activity through alternative pathways. The compound could also increase mitochondrial biogenesis; long-term use might expand oxidative capacity.
Redox balance determines neural resilience.
The GAD Connection: Glutamate and GABA Balance
Glutamate decarboxylase requires pyridoxal-5-phosphate as essential cofactor. This enzyme converts the excitatory neurotransmitter glutamate into the inhibitory GABA; the balance between these systems governs neural excitability. Glutamate-glutamate homeostasis depends partly on B6 availability.
Pyritinol delivers B6 directly to GABAergic neurons; enhanced cofactor availability increases GABA synthesis. This may explain the anxiolytic effects reported by some users; increased inhibitory tone reduces neural hyperexcitability. The mechanism differs from benzodiazepine receptor agonism; pyritinol supports physiological inhibition rather than pharmacological potentiation.
Excitotoxicity protection represents a secondary benefit. Glutamate excess produces neuronal injury through calcium overload; enhanced GABA synthesis and glutamate conversion limit this pathology. Glutamatergic modulation strategies often include B6 support; pyritinol offers targeted delivery.
The GAD67 and GAD65 isoforms differ in cofactor affinity. GAD67 produces baseline GABA for tonic inhibition; GAD65 synthesizes vesicular GABA for phasic release. Pyritinol may differentially support these isoforms; the balance between tonic and phasic inhibition could shift.
GABAergic tone affects cognition and mood. Excessive inhibition produces sedation and cognitive slowing; insufficient inhibition allows anxiety and seizures. Pyritinol’s effects likely fall within the physiological range; subtle optimization rather than dramatic alteration.
Neurotransmitter balance requires precision.
Synergistic Architectures: The Retrieval Accelerator Connection
PRL-8-53 enhances memory retrieval through cholinergic mechanisms distinct from pyritinol’s metabolic approach. Combining these compounds might address multiple cognitive parameters simultaneously; metabolic support plus retrieval acceleration could prove synergistic.
PRL-8-53 improves the threshold of retrieval; information becomes accessible more rapidly. Pyritinol enhances the metabolic substrate for this process; ATP availability supports the ion pumping and neurotransmitter release that retrieval requires. The combination addresses both efficiency and capacity.
Cholinergic synapses are metabolically demanding. Acetylcholine synthesis, packaging, and release consume substantial ATP; vesicular cycling requires continuous energy input. Pyritinol’s metabolic enhancement may support the increased cholinergic activity that PRL-8-53 produces.
Clinical applications might target specific cognitive domains. PRL-8-53 for retrieval-intensive tasks; pyritinol for sustained metabolic support. The combination requires careful titration; individual variation in both compounds necessitates personalized protocols.
Stacking requires scientific rationale.
Pyritinol Risk Assessment and Monitoring Protocol
| Adverse Event | Frequency Estimate | Time to Onset | Clinical Action | Reversibility |
|---|---|---|---|---|
| Stevens-Johnson Syndrome | Rare (<0.1%) | 1-3 weeks | Immediate discontinuation; emergency referral | Variable; potential mortality |
| Pemphigus Vulgaris | Very rare (<0.01%) | 2-8 weeks | Dermatology consultation; immunosuppression | Slow; may persist months |
| Acute Hepatitis | Uncommon (0.1-1%) | 2-12 weeks | Liver function monitoring; discontinuation | Usually complete |
| Mild GI Disturbance | Common (5-10%) | Days | Dose reduction; take with food | Immediate upon cessation |
| Skin Rash (non-SJS) | Uncommon (1-2%) | 1-4 weeks | Discontinue; dermatology if progressive | Complete within weeks |
| Headache | Common (3-8%) | Hours to days | Dose adjustment; hydration | Rapid resolution |
This matrix guides clinical decision-making; individual risk assessment must precede pyritinol prescription. The serious adverse events are rare but potentially fatal; the benign events are common but manageable. Informed consent requires discussion of both categories.
Monitoring protocols should reflect these frequencies. Serious dermatological reactions develop rapidly; patient education ensures prompt recognition. Liver function tests at baseline and periodic intervals detect hepatotoxicity before clinical symptoms; early discontinuation prevents progression.
Risk communication must be clear.
Community Reports: The Legacy Era
The following accounts derive from the early nootropic community; some of these reports date as far back as 10-13 years ago, representing the Legacy Era of cognitive enhancement exploration. While individual experiences vary, these narratives provide context that predates modern regulatory scrutiny.
Clinical Availability and Energetic Effects
Historical availability influenced usage patterns. Some regions permitted over-the-counter purchase; this accessibility enabled broader community experimentation.
Pyritinol is amazing stuff… Available OTC in Mexico as ‘Encephabol 200’. The energy was a bit harsh, like too much caffeine. I discovered it by accident… it improved memory in elderly patients. ; ViolinistBrief6740
The Mexican availability reflects regulatory divergence. Some jurisdictions recognized pyritinol as a pharmaceutical agent; others classified it as a supplement. This inconsistency shaped global access patterns.
Mood and Cognitive Clarity
Subjective effects extend beyond memory enhancement. Users report anxiolytic and mood-elevating properties; these effects may reflect GABAergic mechanisms.
Pyritinol always seems to energise me (without causing the jitters), increase my mood, lessen anxiety without causing drowsiness, felt more confident and in control PLUS it got rid of my brain fog. ; fuckofffibro
The absence of jitters distinguishes pyritinol from stimulants. The effect suggests metabolic optimization rather than sympathetic activation; this aligns with the efficiency hypothesis.
Adverse Reactions and Allergic Responses
Individual tolerance varies dramatically. Some users experience severe reactions; these events highlight the autoimmune risks documented in clinical literature.
Extremely bad reaction after taking 300mg of Pyritinol… blurred vision, uncoordinated and had heart palpitations… lay down and started seeing things with my eyes closed. Another possibility is that I had an allergic reaction… it literally burned my tongue. ; ranca
The burning sensation suggests direct chemical irritation. The disulfide bond may react with mucosal tissues; this local effect predicts systemic hypersensitivity. Visual disturbances and cardiovascular symptoms indicate serious intolerance.
Palatability and Practical Considerations
Subjective experience includes sensory qualities. Taste and texture influence compliance; unpleasant characteristics deter consistent use.
I took it for a while and I guess it was doing something. Also, it tastes like the juice that collects in the bottom of a garbage can. Especially bad, even for nootropics. ; yallapapi
Taste aversion represents a practical barrier. Encapsulation mitigates this issue; enteric coatings further improve tolerability. The sensory characteristics derive from the disulfide chemistry.
Severe Systemic Reactions
The most concerning reports involve multi-system symptoms. Musculoskeletal, autonomic, and neurological effects occurring together suggest serious adverse reactions.
I tried pyrisudanol (mentis) in the past with nefast effects. It was horrible, almost every muscle in my body was aching. Sialorrhea, nausea, blurred vision and sweating together with a nasty permanent headache were added to the pack. ; Dangerous-Jaguar7779
Pyrisudanol represents a related compound with similar disulfide structure. The severe reaction pattern underscores individual variability; genetic factors likely determine susceptibility. These reports informed regulatory restrictions in several markets.
Historical context informs current practice.
Acute Pancreatitis: The Immune-Mediated Danger
Gastroenterology literature documents an additional serious risk associated with pyritinol. Acute pancreatitis appears in adverse event databases; the mechanism involves immune-mediated inflammation rather than direct chemical toxicity. This complication further explains market withdrawals and regulatory caution.
The pancreas synthesizes and secretes digestive enzymes; premature activation within the gland produces autodigestion. Drug-induced pancreatitis typically involves either direct toxic effects or immune-mediated injury. Pyritinol appears to trigger the latter mechanism.
Case reports describe pyritinol-associated pancreatitis developing after variable exposure durations. Some patients experience symptoms within days; others develop pancreatitis after months of uneventful use. This pattern suggests sensitization rather than cumulative toxicity.
The disulfide bond likely serves as the immunogenic trigger. Pancreatic acinar cells express MHC molecules that present drug-modified peptides to T-lymphocytes. Once sensitized, subsequent exposures produce rapid and severe inflammatory responses.
Clinical presentation mirrors other forms of acute pancreatitis. Severe epigastric pain radiates to the back; nausea and vomiting are prominent. Serum amylase and lipase rise markedly; imaging confirms pancreatic inflammation. Severity ranges from mild edematous disease to necrotizing pancreatitis with multi-organ failure.
The immune-mediated mechanism has therapeutic implications. Corticosteroids may attenuate the inflammatory response; discontinuation of pyritinol is mandatory. Supportive care addresses fluid management and nutritional support; severe cases require intensive care.
Genetic predisposition likely influences susceptibility. HLA associations similar to those for dermatological reactions may predict pancreatic involvement. Family history of autoimmune disease or drug reactions should prompt heightened vigilance.
Pancreatitis represents a medical emergency.
The Metabolic Twin: Sulbutiamine Versus Pyritinol
The disulfide modification strategy applies to multiple vitamins. Sulbutiamine represents the thiamine equivalent of pyritinol; two thiamine molecules joined by a disulfide bridge create a lipophilic compound that crosses the blood-brain barrier. These metabolic twins share structural logic but produce distinct clinical effects.
Sulbutiamine primarily enhances physical drive and motivation. Users report increased energy and reduced fatigue; the compound supports peripheral and central thiamine pyrophosphate-dependent enzymes. Asthenia and chronic fatigue respond particularly well; the mechanism involves improved mitochondrial function.
Pyritinol targets cognitive signal processing rather than physical capacity. The compound enhances memory encoding and reduces neural noise; users describe cleaner cognitive function rather than increased energy. This distinction guides clinical selection; physical fatigue favors sulbutiamine while cognitive fog indicates pyritinol.
The choice reflects therapeutic goals. Athletes and physically demanding professions benefit from sulbutiamine; office workers and students typically prefer pyritinol. Some practitioners combine both compounds; this addresses metabolic support comprehensively.
Thiamine pyrophosphate serves critical metabolic functions distinct from B6-dependent reactions. The Krebs cycle and pentose phosphate pathway both require TPP; sulbutiamine enhances these energy-generating processes. Pyritinol supports neurotransmitter synthesis and amino acid metabolism; the effects are more specifically neurological.
Individual response patterns help distinguish appropriate candidates. Those who respond well to thiamine supplementation generally tolerate sulbutiamine; previous B6 responsiveness predicts pyritinol benefit. Genetic variation in vitamin metabolism influences these patterns.
Signal cleaning versus physical drive.
The disulfide strategy revolutionizes vitamin pharmacology.
Future Directions and Research Imperatives
Pyritinol research has stagnated since the 1980s. The compound fell from favor as newer nootropics emerged; modern pharmacology largely ignores this established agent. This represents a missed opportunity; the metabolic mechanisms warrant contemporary investigation.
Advanced imaging could clarify pyritinol’s effects on cerebral metabolism. PET studies tracking glucose utilization would quantify the efficiency improvements suggested by older research. Functional MRI might reveal changes in network connectivity; metabolic enhancement could improve inter-regional coordination.
The autoimmune risk also requires characterization. Modern immunology could identify susceptible individuals; genetic markers might predict adverse reactions. Personalized medicine approaches could restrict pyritinol to appropriate candidates; this would improve the risk-benefit profile.
Pyritinol stands as proof that vitamin derivatives can achieve nootropic effects. The disulfide bridge strategy could apply to other nutrients; similar modifications might enhance central delivery of other polar compounds. The compound deserves renewed attention from researchers and clinicians.


0 Comments