The Molecular Architecture of MAO-B Selectivity
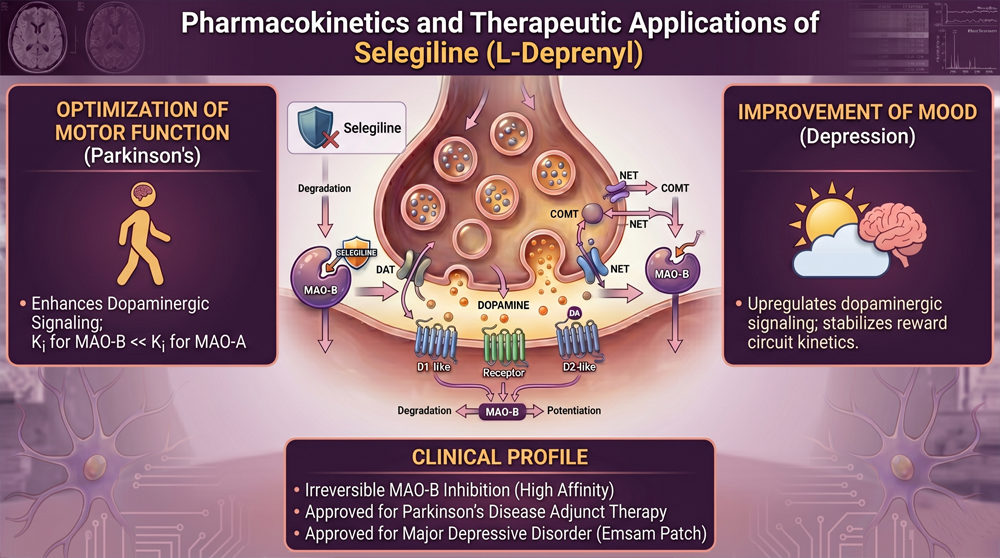
Selegiline contains a propargylamine scaffold; this structure enables irreversible MAO-B inhibition. At low doses of 5mg to 10mg daily, the compound achieves absolute selectivity for the MAO-B isoform, leaving MAO-A activity intact for peripheral metabolic functions.
The propargyl group forms a covalent bond with the enzyme, creating an irreversible adduct that distinguishes selegiline from transient, reversible inhibitors. Recovery of enzyme activity requires the de novo synthesis of new MAO-B proteins, a biological process that typically necessitates a two-week washout period after discontinuation.
Because MAO-B is responsible for the oxidative deamination of dopamine and phenethylamine, blocking this enzyme effectively preserves synaptic neurotransmitter levels in the striatum and prefrontal cortex. This central preference maximizes its utility in neuro-performance contexts without the peripheral side effects associated with broader inhibitors.
The enzyme itself resides primarily in glial cells and serotonergic neurons; its distribution varies significantly across different brain regions. Binding kinetics follow pseudo-first order mechanics, ensuring the covalent adduct forms rapidly upon ingestion.
Beyond MAO Inhibition: The SOD/CAT Induction Protocol
Selegiline upregulates superoxide dismutase gene expression; this antioxidant enzyme neutralizes superoxide radicals before they can initiate cellular damage. Neuroprotective actions extend far beyond simple metabolic inhibition, representing a genuine genomic intervention.
Catalase activity also increases with chronic administration, allowing the cell to decompose hydrogen peroxide into harmless water. This dual antioxidant defense creates a protective cellular environment where oxidative stress is neutralized at the molecular source.
These gene expression changes occur via the activation of specific transcription factor pathways, including NF-kB and AP-1. By increasing anti-apoptotic proteins like Bcl-2, selegiline stabilizes mitochondrial complex I function and maintains cellular energy production capacity under high-stress conditions.
In MPP+ toxicity models, selegiline shields substantia nigra neurons from potent parkinsonian toxins. Hydroxyl radical formation decreases, and iron-catalyzed oxidation reactions are suppressed; this protects the structural integrity of the neuronal membrane and preserves the long-term health of the human machine.
The Knoll Hypothesis: Dopaminergic Preservation and Lifespan Extension
Joseph Knoll originally discovered the longevity effects of selegiline during rat studies where lifespan was extended by approximately thirty percent. The mechanism was attributed to the preservation of the dopaminergic system, as aging rats maintained youthful motor function and vigorous sexual activity into late life.
Rats received chronic low-dose administration; dopamine levels stayed elevated versus controls throughout the aging process. This suggested that maintaining the “dopaminergic engine” was a prerequisite for systemic vitality.
Translating these results to human biology requires a skeptical, systems-analyst perspective. While rats showed sustained neurological vitality on low-dose protocols, human metabolism involves multifactorial aging processes that no single compound can entirely arrest.
Dopaminergic decline is a hallmark of normal human aging. By maintaining tyrosine hydroxylase activity, selegiline ensures that dopamine synthesis capacity resists age-related decay, even if it does not act as a literal fountain of youth.
Metabolic Pathway Engineering and Amphetamine Conversion
First-pass metabolism in the liver converts oral selegiline into three primary metabolites: desmethylselegiline, L-methamphetamine, and L-amphetamine. While these metabolites often carry a stigma, the L-isomer stereoselectivity means they lack the high abuse potential and peripheral jitters associated with D-isomers.
L-methamphetamine has minimal stimulant properties at these concentrations, though it can still contribute to the arousal-promoting effects of the drug. Stimulant side effects including insomnia may emerge; morning dosing is typically required to minimize sleep architecture disruption.
Transdermal delivery via the EMSAM patch offers a way to bypass hepatic first-pass metabolism entirely. This route reduces the formation of amphetamine metabolites by nearly ninety percent, providing a cleaner dopaminergic signal without the systemic stimulant load.
Dopaminergic Resurrection and Iatrogenic Blunting Reversal
SSRI-induced dopaminergic blunting often responds to selegiline because MAO-B blockade allows dopamine to accumulate in the synapse, unmasking reward circuits that have been silenced by chronic serotonin overactivity. This is a primary tool for addressing Post-SSRI Sexual Dysfunction (PSSD) and emotional numbing.
Tyrosine hydroxylase preservation ensures that the synthetic machinery of the brain keeps pace with the metabolic demand. Stacking with L-tyrosine catalyzes substrate availability; synthesis meets metabolic demand without causing a “crash” effect.
Neuroleptic-induced deficits may also improve; dopaminergic signaling resists antipsychotic blockade under certain clinical conditions. The combination requires careful monitoring; theoretical interactions demand clinical vigilance to maintain safety.
Dose-Dependent Selectivity and the Therapeutic Window
Low-dose selegiline maintains absolute MAO-B specificity; 5mg daily achieves substantial enzyme inhibition. The selectivity window is narrow; doses above 20mg compromise MAO-A specificity and introduce new risks.
MAO-A metabolizes serotonin and norepinephrine; inhibiting this enzyme creates dietary restrictions. The “cheese effect” emerges with MAO-A blockade; tyramine can trigger hypertensive crises if left unmonitored.
Clinical dosing exploits this selectivity window; patients receive neuroprotection without dietary constraints. The therapeutic index favors safety; low doses provide technical benefits with minimal systemic risk.
Cerebral MAO-B inhibition parallels plasma levels; enzyme occupancy measures correlate with clinical effects. Positron emission tomography confirms central binding; peripheral inhibition serves as a reliable biomarker for central activity.
SOD and CAT Genomic Upregulation Mechanisms
Superoxide dismutase exists in cytosolic and mitochondrial forms; selegiline increases both isoforms over time. The Cu/Zn-SOD and Mn-SOD genes respond to treatment; transcription rates increase with chronic exposure.
Catalase decomposes hydrogen peroxide into water; this heme-containing enzyme protects cellular structures from internal oxidation. Selegiline administration elevates catalase mRNA; protein synthesis follows this transcriptional activation.
The antioxidant response element drives gene expression; Nrf2 transcription factor binds to ARE sequences. Selegiline activates this pathway; endogenous antioxidant defenses strengthen systematically throughout the neural architecture.
Oxidative stress markers decrease with treatment; lipid peroxidation and protein oxidation diminish significantly. The brain maintains redox balance; neuronal membranes resist oxidative damage even under metabolic stress.
Substantia Nigra Protection and Parkinson’s Applications
Parkinson’s disease involves dopaminergic neurodegeneration; selegiline slows this progression in early stages. The DATATOP trial demonstrated delayed disability; monotherapy postponed the requirement for levodopa by months.
Tyrosine hydroxylase preservation maintains dopamine synthesis; the rate-limiting enzyme remains functional despite pathological pressure. Selegiline protects nigral neurons from apoptosis; mitochondrial function stabilizes under the “Clinical Fortress” environment.
Levodopa pharmacodynamics improve with combination therapy; selegiline extends the dopamine half-life. Motor fluctuations may decrease; on-time increases without a corresponding worsening of dyskinesia.
Clinical Pharmacokinetics and Dosing Protocols
Oral bioavailability is low; extensive first-pass metabolism limits initial absorption. Desmethylselegiline forms rapidly; this active metabolite contributes to the clinical profile of the compound.
The half-life is short for the parent compound; however, metabolites persist for days. Enzyme inhibition outlasts plasma levels; new MAO-B synthesis requires two weeks to return to baseline.
Morning dosing prevents insomnia; the amphetamine metabolites disrupt sleep architecture if taken late in the day. Sublingual administration may accelerate onset; buccal absorption bypasses some hepatic metabolism for a more direct effect.
Safety Profile and Adverse Event Management
Insomnia represents the most common side effect; stimulant metabolites activate arousal pathways. Morning administration minimizes sleep disruption; dose reduction may help resistant cases where sleep quality is compromised.
Orthostatic hypotension occurs occasionally; this reflects autonomic nervous system effects. Gradual dose titration improves tolerance; patients adapt to hemodynamic changes over the first few weeks of treatment.
Serotonin syndrome remains rare; a theoretical risk exists with serotonergic combinations. Clinicians should monitor for agitation; hyperthermia and rigidity require immediate intervention if they manifest.
Dopaminergic Resurrection Protocols: The Selegiline-Bromantane Synergy
Selegiline and Bromantane create a dopaminergic amplification cascade; MAO-B inhibition meets tyrosine hydroxylase upregulation. This combination addresses multiple failure points; dopamine synthesis and metabolic preservation work in concert to restore tone.
Selegiline extends dopamine half-life through metabolic blockade; synaptic dopamine remains available longer. Bromantane increases tyrosine hydroxylase expression; synthetic capacity rises to meet the heightened demand.
The synergy produces sustained dopaminergic tone; acute and chronic mechanisms complement each other. Patients report improved motivation and reward sensitivity; hedonic capacity recovers from iatrogenic blunting induced by years of SSRI use.
L-tyrosine supplementation completes the protocol; substrate availability matches enhanced synthetic capacity. The trio creates a self-reinforcing dopaminergic system; each component supports the structural goals of the others.
The Epigenetic Synergy: SOD/CAT and Gene Expression Preservation
Oxidative stress silences dopaminergic gene expression; free radicals damage DNA and histone structures. Selegiline induces SOD and CAT expression; these enzymes neutralize reactive oxygen species before epigenetic damage can occur.
Tyrosine hydroxylase gene promoters remain accessible; antioxidant protection preserves transcription factor binding. Dopamine synthesis machinery maintains expression; the cellular environment supports continued function even as the patient ages.
Histone acetylation patterns stabilize under reduced oxidative load; chromatin remains in open configurations. Gene silencing through oxidative mechanisms diminishes; dopaminergic neurons retain their technical synthetic capacity.
Mitochondrial DNA also benefits from antioxidant protection; SOD2 specifically guards the mitochondrial genomes. Energy production maintains efficiency; the cellular power grid supports neurotransmitter synthesis without structural brownouts.
PSSD Rescue Context: Unmasking Blunted Reward Circuits
Chronic SSRI exposure suppresses mesolimbic dopamine function; reward circuits become dampened and unresponsive. Selegiline counteracts this through MAO-B inhibition; dopamine accumulates and signaling begins to restore.
The nucleus accumbens regains responsiveness to rewarding stimuli; salient triggers activate appropriate dopamine release once again. Motivation and pleasure-seeking behavior normalize; anhedonia recedes with restored dopaminergic tone.
Prefrontal cortical dopamine improves executive function; decision-making and goal-directed behavior strengthen. The cognitive-emotional integration recovers; affective blunting diminishes systematically through targeted neurotransmitter architecture.
Sexual function particularly benefits from dopaminergic restoration; libido and arousal depend on intact reward processing. Orgasms regain intensity; the pleasure response normalizes as dopamine availability returns to pre-SSRI levels.
Surgical Safety Audit: The Cheese Effect Biochemistry
The cheese effect emerges from tyramine displacement of norepinephrine; MAO-A inhibition prevents norepinephrine breakdown. Hypertensive crisis follows sympathetic overflow; blood pressure rises dangerously in a short period.
At doses below 10mg, selegiline maintains MAO-B selectivity; MAO-A in peripheral tissues remains functional. Dietary tyramine encounters intact MAO-A; norepinephrine breakdown continues normally without the risk of a crisis.
The tyramine molecule structurally resembles catecholamines; displacement of stored norepinephrine occurs readily. MAO-A normally metabolizes released norepinephrine; blockade permits dangerous accumulation that the system cannot handle.
MAO-B inhibition alone does not trigger the cheese effect; norepinephrine metabolism proceeds unaffected. Patients on low-dose selegiline require no dietary restrictions; tyramine sensitivity remains within normal parameters.
Transdermal selegiline complicates this analysis at higher doses; the 12mg patch inhibits MAO-A sufficiently. Dietary restrictions apply at this dosage; the patch bypasses hepatic metabolism but still affects the enzyme threshold.
Selegiline Dosage vs. Enzyme Selectivity Matrix
| Dosage | MAO-B Inhibition | MAO-A Inhibition | Dietary Restrictions |
|---|---|---|---|
| 2.5mg | Moderate (60-70%) | None | None required |
| 5mg | Substantial (80-90%) | Minimal | None required |
| 10mg | Near-complete (>95%) | Minimal | None required |
| 20mg+ | Complete | Moderate | Tyramine restriction advised |
| 40mg+ | Complete | Substantial | Strict MAOI diet required |
Clinical Interactions: Stacking vs. Contraindications
| Combination | Mechanism | Risk Level |
|---|---|---|
| Selegiline + L-Tyrosine | Substrate provision for TH | Low; synergistic |
| Selegiline + Bromantane | Dual dopaminergic enhancement | Low; complementary |
| Selegiline + Mucuna | L-DOPA + metabolic preservation | Moderate; monitor BP |
| Selegiline + SSRI | Theoretical serotonin syndrome | Moderate; clinical caution |
| Selegiline + MAOI | Redundant MAO inhibition | High; contraindicated |
| Selegiline + Stimulants | Additive dopaminergic effects | Moderate; dose adjustment |
Microbiome Interactions and Gut-Brain Modulation
Gut bacteria metabolize selegiline partially; first-pass metabolism begins in intestinal microbiota. Different bacterial profiles alter bioavailability; individual variation reflects microbiome composition and metabolic efficiency.
MAO-B exists in gut tissues; local inhibition affects serotonin and dopamine availability. Gut-derived neurotransmitters influence enteric nervous function; motility and secretion respond accordingly to these shifts.
Probiotic supplementation may optimize selegiline absorption; Lactobacillus and Bifidobacterium strains affect drug metabolism. Prebiotic fiber supports beneficial bacteria; the microbiome-drug interaction warrants further clinical attention.
Chronobiological Optimization and Circadian Factors
MAO-B activity follows circadian rhythms; enzyme levels peak during sleep hours. Morning selegiline dosing aligns with low basal MAO-B; inhibition occurs when enzyme activity naturally diminishes.
Dopaminergic tone requirements vary by time of day; motivation needs peak during waking hours. Selegiline supports daytime function; evening doses disrupt sleep architecture and prevent proper recovery.
Cortisol rhythms interact with dopaminergic signaling; the HPA axis modulation affects mood and energy. Selegiline may blunt cortisol elevation; stress response normalization benefits from this timing optimization.
Neuroplasticity Windows and Recovery Trajectories
Brain-derived neurotrophic factor surges with dopaminergic enhancement; synaptic plasticity increases during treatment windows. Selegiline-induced dopamine elevation triggers BDNF expression; structural remodeling follows molecular changes.
Critical periods for plasticity may exist; early intervention produces superior outcomes. Delayed treatment still yields benefits; the brain retains plasticity capacity throughout the human lifespan.
Recovery from iatrogenic blunting requires sustained intervention; synaptic remodeling needs weeks to months to take hold. Patient expectations must align with biological timelines; gradual improvement characterizes successful treatment.
Biomarker Monitoring and Clinical Assessment
Homovanillic acid in urine reflects dopamine metabolism; 24-hour collections assess metabolic status. Elevated HVA indicates MAO-B inhibition; the biomarker confirms pharmacodynamic effects are taking place.
Platelet MAO-B activity serves as a peripheral proxy; enzyme inhibition parallels central effects. Blood draws measure pharmacological impact; dosing adjustments follow this biomarker feedback.
Subjective scales assess clinical improvement; motivation and pleasure ratings track recovery. Objective measures complement patient reports; targeted evaluation guides treatment optimization over time.
Gender-Specific Considerations and Hormonal Interactions
Estrogen modulates MAO-B expression; women show different enzyme activity patterns throughout their lives. Hormonal fluctuations affect selegiline response; menstrual cycle phase alters baseline dopaminergic tone.
Postmenopausal women may benefit substantially; estrogen decline correlates with dopaminergic dysfunction. Selegiline addresses this deficit; dopaminergic support compensates for these hormonal changes.
Testosterone interacts with dopaminergic signaling; men show distinct response profiles. Selegiline may support androgenic function; dopamine and testosterone systems interconnect bidirectionally within the human machine.
Age-Related Pharmacokinetic Changes
Hepatic metabolism slows with advancing age; CYP450 enzyme activity diminishes. Selegiline clearance decreases in elderly patients; lower doses achieve equivalent effects compared to younger cohorts.
Renal function affects metabolite elimination; desmethylselegiline accumulation occurs with impairment. Dose adjustment prevents toxicity; elderly populations require conservative initiation of any protocol.
Brain MAO-B activity increases with age; the aging brain produces more enzyme over time. Higher selegiline doses may benefit older adults; increased target demands more inhibitor to achieve the same result.
Genetic Polymorphisms and Personalized Dosing
CYP2D6 variants affect selegiline metabolism; poor metabolizers show altered clearance. Extended effects occur in slow metabolizers; dose reduction prevents accumulation and potential side effects.
CYP2C19 also contributes to metabolism; polymorphisms alter metabolic ratios. Individual genetic profiles guide dosing; pharmacogenetic testing optimizes therapy and ensures safety.
MAO-B gene variants affect enzyme levels; BDNF polymorphisms modulate neuroplasticity responses. Personalized medicine approaches incorporate genetics; one size does not fit all in the dopaminergic architect’s world.
Field Intelligence: Clinical Anecdotes and N=1 Log Analysis
Systems architecture requires an audit of real-world metabolic diversion. These community logs provide a technical perspective on how various administration routes alter the pharmacodynamics of the propargylamine scaffold in diverse human machines.
Sublingual Kinetics and HPPD Management
“I have experimented with a lot of substances, but since being diagnosed [with epilepsy] I’ve been stuck trying various supplements. A lot of my brain fog is gone; my HPPD has diminished. I take 3-5mg per day sublingually, which should bypass the metabolic processes that release [L-methamphetamine].”
This report confirms the efficacy of bypassing hepatic first-pass metabolism through sublingual delivery. By avoiding the initial metabolic gateway, the user facilitates the direct systemic entry of the parent compound; this prevents the excessive formation of L-methamphetamine and preserves the pure MAO-B inhibitory signal required for cognitive clarity.
Intranasal Delivery and First-Pass Evasion
“I take selegiline daily; I use it intranasally at around 0.5-1mg. I also take modafinil orally and need selegiline to evade first pass metabolism or I might experience dietary interactions.”
Intranasal administration represents a high-efficiency bypass of the hepatic gateway. Utilizing this route allows for extremely low dosages to maintain high central MAO-B occupancy; the clinical-grade benefit is the preservation of the “Safety Valve” in the gut, which minimizes tyramine sensitivity even when stacking with other dopaminergic agents.
Receptor Affinity and Autoreceptor Isoforms
“Elevation in DA would initially activate high affinity D3 receptors. As you increase the dose you would see the other higher affinity DRs: D2/D4. I believe it can also be an autoreceptor in its short isoform.”
This technical log captures the importance of the therapeutic window. Maintaining low dosages ensures the target activation of specific high-affinity receptor clusters; exceeding these thresholds often triggers compensatory feedback from autoreceptor isoforms, which can paradoxically suppress the very dopaminergic tone the protocol is designed to restore.
Mood Restoration and Executive Drive
“L-deprenyl for longevity… ended up loving it a little too much. Mood improved, increased sex drive, long term brain protection. It felt like it was finally giving me the focus I needed to actually do stuff.”
The restoration of the dopaminergic engine often manifests as a significant increase in executive drive and hedonic capacity. These effects result from the metabolic preservation of endogenous dopamine; the system operates with less friction, allowing for the successful execution of goal-directed behaviors that were previously inhibited by iatrogenic or age-related blunting.
The Stimulant Threshold and Oral Dosing
“Oral dosages beginning from about 7.5mg a day the worst side effect it may produce is insomnia. The mind is powerful; stopping it or increasing the dose are the three main choices.”
There is a clearly defined “Insomnia Threshold” for oral ingestion where metabolic diversion becomes a structural bottleneck. Once the liver converts a significant portion of the dose into amphetamine isomers, the pharmacological profile shifts from pure neuroprotection to systemic arousal; this necessitates morning dosing and strict titration to maintain sleep architecture.
Selegiline Administration Comparison Matrix
| Administration Route | Observed Kinetic Profile | Documented Side Effect | Structural Impact |
|---|---|---|---|
| Oral (5mg – 10mg) | High metabolic diversion | Insomnia; peripheral jitters | Systemic stimulant metabolites. |
| Sublingual (2mg – 5mg) | Direct systemic absorption | Localized irritation (rare) | Pure MAO-B signaling. |
| Intranasal (0.5mg – 1mg) | Maximum hepatic bypass | Sensitive dosing required | Central occupancy at low doses. |
| Transdermal (Patch) | Steady-state maintenance | Site redness; high cost | Superior metabolic bypass. |
The SuperMindHacker Clinical Assessment
Selegiline represents a foundational dopaminergic tool; its mechanisms extend beyond simple enzyme inhibition. The SOD/CAT induction catalyzes genuine neuroprotection; this distinguishes it from mere metabolic blockers.
The Knoll hypothesis intrigues but remains unproven; human longevity applications lack robust evidence. Dopaminergic preservation has clinical merit; Parkinson’s disease applications demonstrate this clearly.
Amphetamine metabolites complicate risk-benefit analysis; transdermal delivery mitigates this concern. The Systems Architect recommends cautious application; evidence guides rational use for every patient.


0 Comments