Guanfacine: Alpha-2A Precision for Executive Function
Guanfacine represents a mechanistically distinct approach to attention deficit hyperactivity disorder. The compound selectively stimulates postsynaptic alpha-2A adrenergic receptors in the prefrontal cortex; this mechanism strengthens network connectivity without global sympathetic activation. Unlike stimulants that flood the synapse with dopamine, guanfacine optimizes signal transmission; the effect is regulatory rather than augmentative.
The prefrontal cortex governs executive function; this region mediates working memory, impulse control, and emotional regulation. Noradrenergic innervation from the locus coeruleus modulates PFC activity; optimal norepinephrine levels enhance signal-to-noise ratios. Too little produces apathy and distractibility; too much generates distractibility through noise. Guanfacine tightens this regulation; it calms the overactive without sedating the appropriately engaged.
Precision beats power.
The Signal-to-Noise Hypothesis: Why More Dopamine Is Not the Answer
Traditional ADHD treatment relies upon dopamine enhancement. Methylphenidate and amphetamine increase catecholamine availability; these drugs improve attention through generalized arousal. The approach works for many patients; however it fails others and produces side effects that limit tolerability.
The limitation of stimulant therapy lies in its mechanism. Increasing dopamine globally enhances both signal and noise; the prefrontal cortex receives more input indiscriminately. Patients experience improved focus alongside anxiety, insomnia, and emotional lability. The therapeutic window is narrow; optimal dosing proves difficult to maintain.
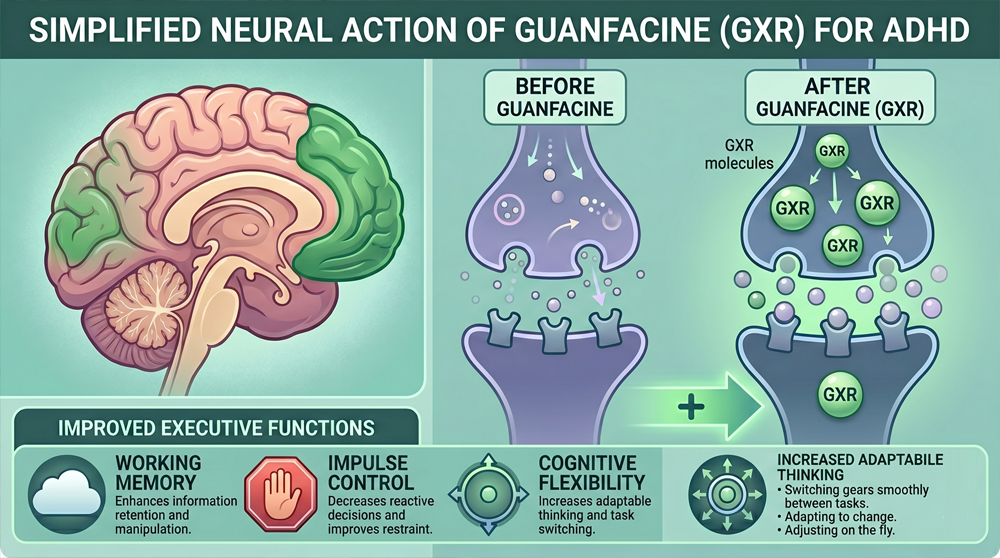
Guanfacine addresses a different bottleneck. The problem in many ADHD cases is not insufficient neurotransmitter but leaky network connectivity. HCN channels on dendritic spines compromise signal transmission; these hyperpolarization-activated cyclic nucleotide-gated channels open inappropriately and shunt synaptic currents. Stimulants cannot repair this structural issue; they merely overwhelm it with excess signal.
The metaphor of leaking pipes fits the physiology. Adding more water pressure to a leaky system wastes resources; fixing the leaks addresses the root cause. Guanfacine closes HCN channels through cAMP inhibition; network connectivity strengthens without increased transmitter release. The effect is more efficient cognition rather than merely more energetic cognition.
Efficiency enables sustainability.
Alpha-2A Selectivity: The Guanfacine Advantage
Alpha-2 adrenergic receptors exist in multiple subtypes; alpha-2A, alpha-2B, and alpha-2C mediate distinct physiological effects. The alpha-2A subtype predominates in the prefrontal cortex; stimulation here enhances working memory and attention. Other subtypes regulate peripheral vascular tone and sedation; their activation produces unwanted effects.
Guanfacine shows selectivity for alpha-2A over other subtypes; this profile maximizes cognitive benefits while minimizing side effects. The compound enhances PFC function at doses that spare peripheral alpha-2 receptors; blood pressure changes are modest and sedation is less pronounced than with non-selective alternatives.
Clonidine illustrates the importance of selectivity. This older alpha-2 agonist lacks subtype discrimination; it activates all three subtypes with roughly equivalent affinity. The result is effective blood pressure reduction but problematic sedation; patients struggle with fatigue and somnolence that limit compliance. Guanfacine’s selectivity profile addresses these limitations; it critical cognitive enhancement without the heavy sedative burden.
The clinical distinction is meaningful. Clonidine requires bedtime dosing to manage sedation; this limits its utility for daytime symptom control. Guanfacine can be administered in the morning; the cognitive benefits persist through waking hours without overwhelming somnolence. Some patients require divided dosing; the extended-release formulation maintains stable levels throughout the day.
Selectivity determines tolerability.
The Executive Function Matrix: Working Memory, Impulse Control, Emotional Regulation
Executive function comprises multiple distinct capacities; guanfacine affects each through common mechanisms. Working memory relies upon sustained delay-period activity in the prefrontal cortex; neurons must maintain firing across seconds without external input. HCN channel closure strengthens this persistent activity; information remains accessible for manipulation.
Impulse control benefits from enhanced top-down regulation. The prefrontal cortex inhibits subcortical structures that generate immediate responses; strengthened connectivity improves this inhibitory control. Patients report reduced impulsive speech and action; the delay between impulse and execution lengthens sufficiently for consideration.
Emotional regulation represents a less recognized but equally important effect. The prefrontal cortex modulates amygdala responses to emotional stimuli; strengthened connectivity reduces emotional lability. Patients with ADHD often experience rejection sensitivity and mood instability; guanfacine addresses these symptoms through improved cortical control rather than direct mood elevation.
The three domains interact in daily functioning. Working memory maintains task goals; impulse control prevents diversion; emotional regulation manages frustration when obstacles arise. Guanfacine’s broad effects on PFC connectivity support this integrated executive function; the benefits extend beyond simple attention enhancement.
Integration produces functional improvement.
Circuit-Level Mechanisms: HCN Channels and cAMP Modulation
The molecular mechanism of guanfacine involves inhibition of adenylate cyclase. Alpha-2A receptor activation reduces cAMP production; this second messenger mediates multiple cellular processes. The effect on HCN channels is particularly relevant; cAMP binding gates these channels, so reduced cAMP promotes channel closure.
Closed HCN channels increase membrane resistance; synaptic currents produce larger voltage changes. The effect is stronger dendritic integration; inputs from multiple synapses sum more effectively. Network connectivity strengthens because signals propagate more reliably; the PFC functions as a more coherent system.
The mechanism differs fundamentally from stimulant action. Dopamine and norepinephrine reuptake inhibitors increase transmitter availability; this enhances signaling through conventional synaptic transmission. Guanfacine modifies the intrinsic properties of postsynaptic neurons; it changes how inputs are processed rather than merely increasing input quantity.
This distinction explains the different side effect profiles. Stimulants affect reward circuits broadly; they produce euphoria and have abuse potential. Guanfacine’s action is confined to cortical regulatory circuits; the compound lacks recreational appeal and shows minimal abuse liability. The FDA scheduling reflects this difference; guanfacine is non-controlled while stimulants are Schedule II.
Mechanism determines risk profile.
Clinical Pharmacology and Dosing Considerations
Guanfacine demonstrates dose-dependent effects that require careful titration. Lower doses primarily enhance prefrontal function; higher doses activate additional receptor populations and produce sedation. The therapeutic window is wide but not infinite; individual optimization is critical.
Extended-release formulations permit once-daily dosing; this improves compliance compared to immediate-release versions. Steady-state levels avoid peaks that produce side effects; the cognitive benefits persist throughout the dosing interval. Morning administration is typical; some patients benefit from evening doses if sedation is manageable.
Combination with stimulants represents common practice. Guanfacine addresses the PFC connectivity that stimulants cannot repair; the combination produces additive or synergistic benefits. Patients who respond partially to either drug alone often show improved outcomes with both. The combination requires careful monitoring; blood pressure and sedation require attention.
Tapering is critical when discontinuing. Receptor upregulation during chronic treatment produces rebound hypertension if stopped abruptly; gradual reduction over weeks prevents this complication. Patient education about proper discontinuation is mandatory; sudden cessation can produce serious cardiovascular effects.
Dosing requires precision.
Adverse Effects and Clinical Management
Sedation represents the most common limiting side effect. The same alpha-2A activation that enhances PFC function can produce somnolence when excessive; dose reduction or divided dosing addresses this. The sedative effect often diminishes with continued treatment; tolerance develops to some degree.
Hypotension and bradycardia reflect peripheral alpha-2 receptor activation. These effects are generally modest at therapeutic doses; however they can be problematic in patients with baseline cardiovascular issues. critical sign monitoring is prudent during dose escalation; significant reductions require dose adjustment.
Dry mouth and constipation result from autonomic effects. These are manageable through symptomatic treatment; they rarely require discontinuation. The side effect profile is generally favorable compared to stimulants; the absence of growth suppression and insomnia is particularly relevant for pediatric patients.
Withdrawal symptoms merit attention. Rebound hypertension can occur with abrupt discontinuation; patients must be counseled about proper tapering. Anxiety and irritability may emerge during withdrawal; these reflect loss of the regulatory effects that the drug provided.
Management minimizes harm.
Synergistic Architectures: Combining with Metabolic and Vascular Support
Guanfacine’s mechanism suggests productive combinations with other cognitive enhancers. The compound optimizes prefrontal connectivity; metabolic and vascular support compounds can provide the substrate that optimized networks require. Rational stacking addresses multiple levels of cognitive function simultaneously.
Pyritinol critical metabolic substrate for the enhanced PFC activity that guanfacine produces. The B6 derivative enhances cerebral glucose utilization; more ATP becomes available for the ion pumping and neurotransmitter release that strengthened networks demand. The combination addresses both infrastructure and energy supply; guanfacine fixes the pipes while pyritinol ensures adequate water pressure.
Hydergine offers complementary adrenergic modulation through different mechanisms. This ergot-derived compound also affects alpha-adrenergic receptors; however its vascular and metabolic effects differ from guanfacine’s direct PFC action. The combination might enhance cerebral perfusion while guanfacine optimizes network connectivity; vascular delivery and synaptic efficiency would improve together.
Stimulant combinations represent established clinical practice rather than theoretical speculation. Guanfacine plus methylphenidate or amphetamine addresses both the dopaminergic and noradrenergic contributions to attention; the mechanisms are complementary. The combination often permits lower stimulant doses; side effects diminish while efficacy improves.
Individual response guides stacking decisions. Some patients show clear guanfacine monotherapy response; others require combination approaches. Systematic trials of single agents before combinations establish contribution attribution; this methodical approach optimizes outcomes.
Rational combinations maximize benefit.
Pediatric Considerations and Developmental Effects
Guanfacine holds particular advantages for pediatric ADHD treatment. The absence of growth suppression distinguishes it from stimulants; this concern troubles many parents considering pharmacotherapy. The non-controlled status simplifies prescribing; refills do not require monthly visits.
Developmental effects require long-term consideration. The prefrontal cortex continues maturing into the mid-twenties; interventions during development may have lasting effects. Whether guanfacine enhances normal development or merely compensates for dysfunction remains unclear; research into these questions is ongoing.
The mechanism suggests possible benefits for academic performance beyond symptom control. Working memory and impulse control contribute to learning; guanfacine’s effects on these domains may improve educational outcomes. However cognitive enhancement in healthy children raises ethical concerns; the boundary between treatment and enhancement blurs.
Clinical judgment must guide pediatric use.
The Binding Affinity Landscape: Alpha-2A Surgical Precision
Guanfacine demonstrates remarkable selectivity for the alpha-2A adrenergic receptor subtype over alpha-2B and alpha-2C variants. The binding affinity ratio defines the therapeutic window; high selectivity maximizes prefrontal cortical effects while minimizing peripheral and sedative actions. This pharmacological precision distinguishes guanfacine from non-selective alternatives.
Ki values reveal the molecular basis of selectivity. Guanfacine shows approximately 15-fold higher affinity for alpha-2A compared to alpha-2B receptors; the alpha-2C distinction is similarly pronounced. Clonidine lacks this discrimination; it binds all three subtypes with roughly equivalent affinity. The numerical difference translates directly into clinical experience.
The alpha-2A subtype predominates in the prefrontal cortex; this distribution explains guanfacine’s cognitive benefits. Alpha-2B receptors regulate vascular smooth muscle; their activation produces hypotension. Alpha-2C receptors mediate sedation and thermoregulation; stimulation here generates somnolence. Guanfacine’s selectivity profile targets the therapeutic receptor while sparing the problematic ones.
Ratios determine outcomes.
The Surgical Shield Against Sedation
Selectivity ratios function as a pharmacological shield. The 15-fold preference for alpha-2A creates a therapeutic window where cognitive enhancement occurs at doses below those producing significant alpha-2B/2C activation. This shield permits daytime dosing; patients function without overwhelming somnolence.
The concept mirrors surgical precision. Non-selective alpha-2 agonists are blunt instruments; they improve cognition but disable the patient through sedation. Guanfacine operates like a specialized tool; it addresses the specific pathology without collateral damage. The surgical shield is the selectivity ratio; it protects against off-target effects.
Clinical dosing exploits this shield. Extended-release formulations maintain concentrations within the selective range; peak levels stay below thresholds for significant alpha-2B/2C activation. Immediate-release versions produce higher peaks; the selectivity advantage diminishes at supratherapeutic concentrations. Formulation choice matters as much as compound choice.
Precision enables function.
Pharmacokinetic and Pharmacodynamic Dynamics
The absorption and distribution of guanfacine differ dramatically between immediate-release and extended-release formulations. Understanding these differences guides clinical decision-making; formulation selection affects both efficacy and tolerability.
Immediate-release guanfacine reaches peak plasma concentration within two to four hours; the half-life is approximately 17 hours. This prolonged elimination permits once-daily dosing despite rapid absorption; however the peak-to-trough ratio generates variability in receptor occupancy. Patients experience higher effects shortly after dosing; this temporal pattern can produce sedation during peak exposure.
Extended-release formulations employ an osmotic delivery system; drug release occurs gradually over the dosing interval. Peak concentrations are lower and delayed; the steady-state profile reduces receptor occupancy fluctuations. The pharmacokinetic smoothness translates into improved tolerability; patients report less peak-related sedation while maintaining cognitive benefits throughout the day.
Steady states produce stable effects.
Cmax and Therapeutic Window Optimization
Maximum plasma concentration determines receptor activation intensity. Extended-release guanfacine achieves Cmax values approximately 60% lower than equivalent doses of immediate-release formulations; this reduction stays within the alpha-2A selective range. The lower peaks avoid alpha-2B/2C activation; the surgical shield remains intact.
The therapeutic window is dose-dependent. At 1 milligram daily, extended-release guanfacine produces selective alpha-2A effects in most patients; at 4 milligrams, even the extended formulation may activate additional subtypes. Individual variation in metabolism and receptor density affects this window; some patients require dose reduction to maintain selectivity.
Food effects differ between formulations. Immediate-release guanfacine absorption increases with high-fat meals; this interaction can push concentrations beyond the selective range. Extended-release formulations show minimal food effect; the osmotic system controls release regardless of gastric contents. Consistent pharmacokinetics support predictable clinical effects.
Formulation shapes experience.
G-Protein Coupling and Intracellular Cascade
Alpha-2A receptors couple to Gi/o proteins; this classification determines the intracellular signaling cascade. Agonist binding triggers GDP-GTP exchange on the G-protein alpha subunit; the activated complex dissociates from the receptor and modulates downstream effectors. The specific pathway explains guanfacine’s mechanism.
The primary target is adenylate cyclase. Gi/o activation inhibits this enzyme; cAMP production decreases within seconds of receptor stimulation. Reduced cAMP has multiple consequences; protein kinase A activation diminishes, gene transcription patterns shift, and ion channel gating changes. The HCN channel effect is particularly relevant to cognitive function.
HCN channels require cAMP for normal operation. These hyperpolarization-activated cyclic nucleotide-gated channels open in response to membrane hyperpolarization; cAMP binding shifts the activation curve toward depolarized potentials. Reduced cAMP makes the channels less responsive; they open less frequently and close more rapidly. The effect on dendritic spines is profound.
Closure strengthens networks.
From Molecular Event to Cognitive Enhancement
The molecular cascade culminates in improved prefrontal network function. Closed HCN channels increase membrane resistance; synaptic currents produce larger voltage changes. Dendritic integration strengthens because inputs sum more effectively; the signal reaches the axon hillock with greater fidelity.
This biophysical change manifests as improved working memory. Neurons in the prefrontal cortex must maintain persistent activity during delay periods; this sustained firing represents the neural basis of working memory. HCN channels normally limit this persistence by shunting currents; guanfacine-induced closure removes this brake. Information remains active across longer intervals.
The temporal dynamics matter. G-protein activation occurs within milliseconds of agonist binding; however the cognitive effects develop over days of chronic treatment. Receptor desensitization and downstream adaptations likely contribute; the acute and chronic mechanisms may differ. Chronic guanfacine treatment produces lasting changes in PFC function that persist beyond the drug’s half-life.
Time transforms molecules into cognition.
Guanfacine Technical Specifications
| Parameter | Guanfacine ER | Guanfacine IR | Clinical Implication |
|---|---|---|---|
| Alpha-2A Ki (nM) | ~3.5 | ~3.5 | High affinity enables low-dose efficacy |
| Alpha-2B/A Selectivity | 15:1 (inherent) | 15:1 (inherent) | Surgical shield against sedation |
| Tmax (hours) | 6-8 | 2-4 | ER delays peak; smoother profile |
| Half-Life (hours) | 16-18 | 16-18 | Supports once-daily dosing |
| Cmax (ng/mL per mg) | ~1.2 | ~2.0 | ER achieves 40% lower peaks |
| Metabolism | Hepatic CYP3A4 | Hepatic CYP3A4 | Drug interactions require attention |
This table guides clinical selection between formulations. The identical receptor affinity indicates equivalent mechanism; the pharmacokinetic differences determine tolerability. Extended-release is preferred for most patients; immediate-release may suit those requiring dose flexibility or those with specific metabolic considerations.
Individual variation affects all parameters. CYP3A4 polymorphisms alter clearance; extensive metabolizers may require higher doses. Body weight and composition influence distribution; pediatric dosing requires mg/kg conversion. The specifications represent population averages; individual optimization remains critical.
Data inform practice.
Comparative Pharmacology: Guanfacine vs. Alternatives
| Feature | Guanfacine | Clonidine | Methylphenidate |
|---|---|---|---|
| Primary Mechanism | Alpha-2A selective agonist | Non-selective alpha-2 agonist | Dopamine/norepinephrine reuptake inhibitor |
| Selectivity Ratio | 15:1 (2A:2B) | ~1:1 (non-selective) | Indirect; no alpha-2 action |
| Sedation Profile | Mild (dose-dependent) | Moderate to severe | Minimal (activating) |
| Primary Effect Site | Postsynaptic PFC | Presynaptic autoreceptors | Synaptic cleft (dopamine) |
| Schedule Status | Non-controlled | Non-controlled | Schedule II |
The comparison reveals mechanistic diversity in ADHD treatment. Guanfacine and clonidine share the alpha-2 agonist classification but differ profoundly in selectivity; guanfacine’s precision produces superior tolerability. Methylphenidate operates through entirely different mechanisms; the combination of these approaches often produces synergistic benefits.
Clinical selection depends upon patient characteristics. Those with prominent hyperactivity may prefer methylphenidate; patients with anxiety or sleep disturbance may tolerate guanfacine better. The non-controlled status of guanfacine simplifies prescribing; this practical consideration influences treatment selection independent of pharmacology.
Choice requires individualized assessment.
Network Connectivity and Complementary Approaches
The prefrontal cortex does not function in isolation. Connections to subcortical structures and other cortical regions determine executive function; guanfacine’s effects propagate through these networks. Complementary compounds that support other nodes in these circuits may enhance overall outcomes.
Stimulant medications remain the first-line treatment for most ADHD presentations. Methylphenidate and amphetamine combinations address dopaminergic transmission; guanfacine complements this through noradrenergic modulation. The combination often permits dose reduction of both agents; improved efficacy with reduced side effects represents the therapeutic ideal.
Non-stimulant alternatives offer options for those who cannot tolerate or do not respond to dopaminergic agents. Alpha-GPC critical cholinergic support; this mechanism complements guanfacine’s noradrenergic action. The combination addresses multiple neurotransmitter systems; broader pharmacological coverage may benefit complex cases.
Individual response guides selection. Some patients show robust response to single-agent therapy; others require combinations to achieve functional improvement. Systematic trials establish the minimal effective regimen; polypharmacy should be justified by clear benefit.
Optimization requires patience.
Clinical Anecdotes: The Human Experience
Subjective reports illuminate pharmacological effects that objective measures cannot capture. The following accounts derive from clinical users; they illustrate the practical reality of guanfacine treatment beyond trial data.
The Executive Silence
Many patients describe the onset of guanfacine effects as a qualitative shift in mental experience. The compound does not produce euphoria or obvious stimulation; instead it creates a background of improved regulatory function. Users report that cognitive control becomes accessible rather than effortful.
It was honestly the best thing for my ADHD; it felt like a ‘quiet’ in my brain that I never had before. Instead of five different thoughts screaming at once, there was just one. The executive control and ability to just ‘start’ a task was night and day. ; u/unnamedpfc
The metaphor of mental quiet suggests reduced neural noise. The prefrontal cortex achieves more efficient processing; competing inputs no longer overwhelm executive resources. This subjective experience aligns with the HCN channel mechanism; closure of these leak channels reduces background conductance.
Task initiation improves because attentional resources become deployable. Previously the effort to start activities faced resistance from competing stimuli; guanfacine reduces this competition. The executive silence enables action rather than contemplation.
Quiet enables focus.
The Blood Pressure Adjustment
Cardiovascular effects represent the most common practical challenge with guanfacine initiation. The alpha-2A selectivity does not completely spare peripheral receptors; vascular tone adjustments occur particularly during dose titration. Patients must accommodate these physiological changes.
It basically slows down everything in your brain. I’ve found it incredible for Rejection Sensitive Dysphoria (RSD); nothing feels like a personal attack anymore. However; you have to be careful with the adjustment. The blood pressure drop made me dizzy for the first two weeks before my system stabilized. ; u/eggy1
The reference to rejection sensitive dysphoria highlights an underrecognized guanfacine effect. Emotional regulation improves through strengthened prefrontal-amygdala connectivity; perceived social threats lose their sting. This benefit often emerges before cognitive improvements become obvious.
The cardiovascular adjustment period requires patience. Alpha-adrenergic receptors in vascular smooth muscle downregulate over time; blood pressure stabilizes as compensatory mechanisms engage. Gradual dose titration minimizes these symptoms; aggressive dosing produces more pronounced adjustment challenges.
Adaptation requires time.
The Robotic Wall
Dose optimization presents genuine clinical challenges. The therapeutic window varies between individuals; some patients experience blunting at doses that others tolerate without issue. Finding the optimal dose requires careful titration and attention to subjective experience.
Emotional blunting is very real on this molecule at higher doses. I reached 4mg and felt like I lost my personality. I was productive, sure, but I didn’t care about anything. I felt like a robot going through the motions until I titrated back down to a 1mg dose. ; u/LushLife77
The dose-response relationship for guanfacine is nonlinear. Lower doses produce selective alpha-2A effects that enhance cognition; higher doses activate additional receptor populations that may produce apathy. The 4-milligram experience represents supratherapeutic dosing for this individual; the therapeutic window lay at lower concentrations.
The robotic sensation suggests excessive noradrenergic tone. Prefrontal function follows an inverted U-shaped curve; optimal arousal produces best performance while excessive arousal impairs function. Guanfacine dose must target the ascending limb of this curve; higher doses may push patients onto the descending limb.
Individualization prevents blunting.
SuperMindHacker Guanfacine Implementation Matrix
| Executive Target | Primary Mechanism | Dosing Protocol | Synergistic Stack |
|---|---|---|---|
| Impulse Control | HCN Channel Closure | 1mg PM Titration | Magnesium L-Threonate |
| Emotional Resilience | Alpha-2A Agonism | 2mg Extended Release | Pyritinol (Metabolic Support) |
| Working Memory | Postsynaptic PFC Potentiation | 1mg AM/PM Split | Hydergine (Vascular Synergy) |
This matrix critical a framework for clinical implementation; individual requirements vary. Starting with the lowest effective dose minimizes side effects; titration based on response optimizes outcomes. The synergistic stacks address complementary mechanisms; combination approaches often outperform monotherapy.
Magnesium L-threonate supports NMDA receptor function; this complements guanfacine’s HCN channel effects on dendritic integration. Pyritinol enhances metabolic substrate availability; emotional regulation requires energy-intensive prefrontal processing. Hydergine improves cerebral perfusion; working memory benefits from enhanced vascular delivery.
Personalization maximizes benefit.
Clinical Synthesis and Practical Application
Guanfacine represents a precision tool for executive function enhancement. The compound does not globally activate the brain; it specifically strengthens prefrontal cortical networks through alpha-2A selective mechanisms. This specificity produces distinct benefits and limitations compared to alternative approaches.
Patient selection should consider the mechanism. Those with prominent prefrontal dysfunction; working memory deficits, impulse control problems, and emotional dysregulation show clearest response. Pure hyperactivity without executive impairment may respond better to stimulants; the optimal match depends upon individual pathophysiology.
Dose optimization requires patience and attention to subjective experience. The therapeutic window varies; some patients achieve optimal effects at 1 milligram while others require 3 or 4 milligrams. Signs of blunting or excessive sedation indicate supratherapeutic dosing; reduction restores function.
The future of executive function treatment lies in precision pharmacology. Guanfacine exemplifies this approach; receptor subtype selectivity enables targeted intervention. As biomarker development advances; clinicians will match specific receptor profiles to optimal compounds. The era of trial-and-error psychopharmacology will yield to personalized neurotherapeutics.
Science guides practice.
Future Directions and Research Imperatives
Guanfacine’s mechanism suggests applications beyond ADHD. Prefrontal dysfunction contributes to numerous psychiatric conditions; traumatic brain injury, schizophrenia, and mood disorders all involve PFC impairment. Clinical trials in these populations are warranted; the safety profile supports exploratory investigation.
The alpha-2A selective approach may inform drug development. Other compounds with similar selectivity profiles could offer improved pharmacokinetics or reduced side effects. The molecular target is validated; medicinal chemistry efforts could produce next-generation agents.
Biomarker development would enhance precision prescribing. Identifying patients with HCN channel-mediated PFC dysfunction would guide guanfacine selection; those with primarily dopaminergic deficits might respond better to stimulants. Neuroimaging or genetic markers could stratify treatment; personalized medicine would replace trial-and-error.
Research must advance.
Guanfacine represents a mechanistically sophisticated approach to cognitive enhancement. The compound does not merely increase neurotransmission; it repairs the network infrastructure that enables effective cognition. This precision distinguishes it from older therapies; it points toward a future of targeted neurological intervention.
Understanding precedes mastery.


0 Comments