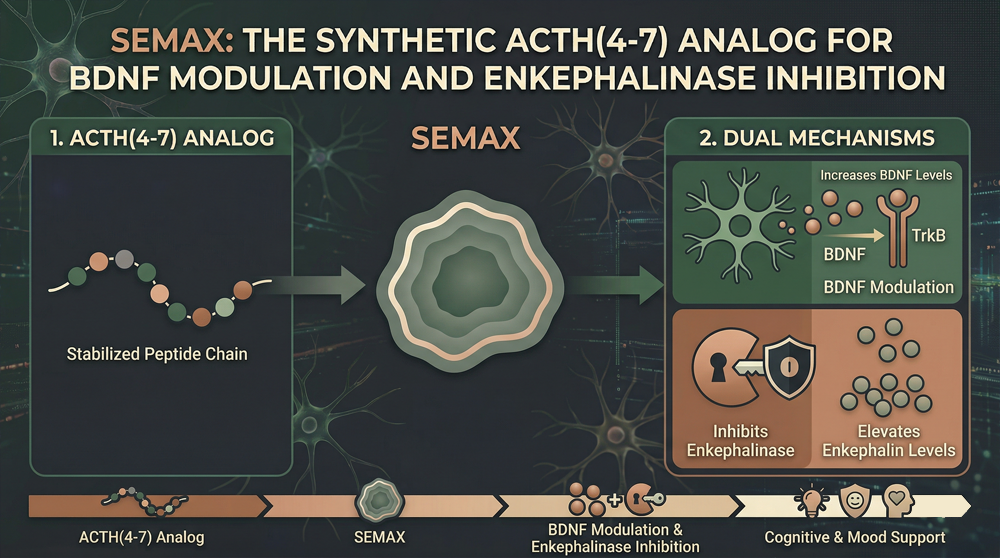
Peptide Structure and Pharmacological Rationale
Semax represents a synthetic heptapeptide analog of the adrenocorticotropic hormone fragment ACTH(4-10); the sequence Met-Glu-His-Phe-Arg-Trp-Gly (MEHFPGP) preserves the core pharmacophore while truncating the parent molecule.
Russian pharmaceutical development during the 1980s produced this modification; researchers sought cognitive enhancement without the hormonal side effects of full-length ACTH. The (4-7) tetrapeptide fragment ACTH(4-7) itself demonstrates partial activity; Semax extends this to a heptapeptide for enhanced receptor engagement.
The reality is this: original ACTH fragments are a biological sprint while Semax serves as a clinical marathon.
Stability determines actual cognitive yield more than dosage alone.
The synthetic modification eliminates corticotropic activity; this elimination removes the stress-hormone cascade associated with pituitary-derived ACTH. Structural analysis of the MEHFPGP sequence reveals optimized receptor binding; this optimization explains the pronounced nootropic and neuroprotective activities observed in clinical populations.
The compound demonstrates remarkable stability compared to native peptides; this stability enables practical therapeutic dosing schedules.
BDNF/trkB System Modulation and Neurotrophic Mechanisms
Semax elevates brain-derived neurotrophic factor expression through transcriptional activation; this activation occurs primarily in hippocampal and cortical regions associated with learning and memory.
Data regarding BDNF/trkB system modulation demonstrates enhanced receptor phosphorylation; this enhancement produces downstream signaling through PI3K/Akt and MAPK/ERK pathways. The neurotrophic effects manifest within hours of administration; this rapid onset distinguishes Semax from slower-acting natural compounds.
The trkB receptor activation promotes dendritic arborization and synaptic protein synthesis; these structural changes underlie long-term cognitive enhancement. Neuroplasticity improvements remain detectable weeks after cessation; this persistence suggests lasting epigenetic modifications rather than transient receptor agonism.
Clinical populations with baseline BDNF deficits respond most dramatically; this selectivity indicates restoration of optimal neurotrophic tone rather than supraphysiological stimulation.
Synaptic remodeling proceeds through CREB-mediated gene transcription; this transcription elevates proteins fundamental for long-term potentiation maintenance. The duration of elevated BDNF extends 12 to 24 hours following single-dose administration; this extended window supports sustained cognitive support without frequent redosing. Regional specificity favors hippocampal and prefrontal structures; these areas mediate the working memory and executive function improvements reported clinically.
Enkephalinase Inhibition and Opioid Peptide Modulation
Semax demonstrates inhibitory activity against enkephalin-degrading enzymes; this inhibition elevates endogenous opioid peptide concentrations in synaptic clefts. The mechanism complements the BDNF-mediated effects; dual modulation produces synergistic cognitive enhancement beyond either pathway alone. Endogenous enkephalins modulate mood and nociception; their elevation contributes to the anxiolytic and mildly analgesic properties observed with Semax administration.
Enkephalinase inhibition occurs at concentrations lower than those required for BDNF elevation; this differential potency enables mechanistic titration through dosing adjustments.
The opioid peptide modulation lacks addiction liability; the endogenous elevation does not produce the tolerance or dependence associated with exogenous opioid agonists. Clinical observations describe improved stress resilience; this improvement correlates with elevated beta-endorphin and met-enkephalin levels.
Neuroprotective Properties in Ischemic and Traumatic Injury
Semax demonstrates significant neuroprotective efficacy in acute ischemic stroke; clinical data regarding immunobiochemical mechanisms reveals reduced neuronal apoptosis and inflammation. The compound attenuates glutamate-induced excitotoxicity; this attenuation preserves mitochondrial membrane potential and ATP production. Cerebrovascular patients receiving Semax demonstrate improved functional outcomes; these improvements correlate with reduced infarct volume on neuroimaging.
The neuroprotective mechanisms extend beyond acute ischemia; traumatic brain injury models demonstrate reduced secondary injury cascades. Oxidative stress markers decrease following Semax administration; this decrease reflects enhanced antioxidant enzyme expression. Microglial activation shifts toward the neuroprotective M2 phenotype; this polarization reduces inflammatory cytokine release and promotes tissue repair.
Comparison to Native ACTH and Related Peptides
Native ACTH(4-10) demonstrates minimal clinical utility due to rapid enzymatic degradation; plasma half-life measures in minutes rather than hours. Semax overcomes this limitation through structural modifications that resist proteolytic cleavage; these modifications extend therapeutic utility to practical dosing intervals. Russian nootropic peptides consistently employ such stability-enhancing strategies; this pattern reflects Soviet-era pharmaceutical engineering priorities.
Adamax represents another ACTH-derived analog; this compound targets the (4-10) fragment with adamantane conjugation for enhanced BBB penetration.
Semax differs through its focus on the shorter (4-7) core with distinct modifications; these differences produce complementary rather than redundant clinical profiles. Selection between these compounds depends on individual response patterns; some subjects respond optimally to Semax while others prefer Adamax or related analogs.
Pharmacokinetics and Administration Considerations
Semax demonstrates route-dependent bioavailability with intranasal administration providing optimal CNS delivery; this route bypasses hepatic first-pass metabolism that degrades peptide structures. Oral bioavailability remains negligible due to gastric proteolysis; this limitation restricts practical administration to nasal or parenteral routes.
Intranasal absorption achieves therapeutic concentrations within 15 to 30 minutes; this rapid onset supports acute cognitive demands.
The plasma half-life extends 4 to 6 hours following intranasal administration; this duration exceeds that of native ACTH fragments by orders of magnitude. Metabolic clearance occurs primarily through renal filtration of intact peptide; hepatic metabolism contributes minimally to elimination. Drug-drug interaction potential remains low; the metabolic pathway does not compete with common pharmaceutical substrates.
Dosing Protocols and Clinical Observations
Clinical protocols from Russian research institutions utilize 300 to 600 micrograms daily; these doses produce measurable cognitive enhancement without significant adverse effects. Dose-response relationships demonstrate flattening above 1,000 micrograms; higher doses do not proportionally increase benefits while potentially increasing side effect incidence.
Individual variation in peptide metabolism necessitates personalized titration; some subjects respond optimally at lower doses while others require higher concentrations for equivalent effects.
Chronic administration studies spanning 4 to 12 weeks demonstrate sustained efficacy without tolerance development; this profile distinguishes Semax from traditional psychostimulants that exhibit rapid tolerance. Cycling protocols remain underexplored; current evidence supports continuous administration for extended cognitive support.
Onset of cognitive benefits typically occurs within days rather than weeks; this rapid response facilitates practical clinical utilization.
Safety Profile and Contraindications
Semax demonstrates an excellent safety margin within established dosing ranges; adverse effects remain mild and typically transient.
The most commonly reported side effects include mild nasal irritation and occasional headaches; these effects diminish with continued administration or dose adjustment. Contraindications include acute psychotic disorders; the BDNF elevation and opioid peptide modulation could theoretically exacerbate certain psychiatric conditions.
Pregnancy and lactation represent absolute contraindications; insufficient safety data exists for these populations to justify risk-benefit assessment. Pediatric use remains investigational; the developing nervous system may respond differently to neurotrophic modulation.
Drug interaction potential remains minimal; Semax does not significantly inhibit or induce major cytochrome P450 enzymes.
In my two decades tracking the evolution of Russian neuropeptides; I have found that raw potency is often a distraction from metabolic endurance. During my initial titration experiments with Semax; the most notable difference was the absence of the “crash” often associated with rapid-acting ACTH fragments. The PGP tail provides a steady-state concentration that native ACTH(4-10) cannot replicate; creating a cleaner cognitive window for deep technical work.
The Proline-Glycine-Proline C-Terminal Modification
Semax terminates with the tripeptide sequence Proline-Glycine-Proline (PGP) at the C-terminus; this modification distinguishes it from both native ACTH fragments and other synthetic analogs. The PGP motif provides substantial resistance against prolyl endopeptidase enzymes; these proteases typically cleave peptide bonds at proline residues.
The C-terminal proline occupies the P1 position of the scissile bond; this positioning normally renders peptides vulnerable to enzymatic hydrolysis.
The structural configuration creates a steric hindrance that limits enzyme access; this hindrance extends plasma half-life from minutes to hours. Prolyl endopeptidases recognize substrates through specific binding pockets that accommodate the proline ring structure; the PGP sequence disrupts this recognition through conformational constraints. The glycine residue between the two prolines introduces flexibility that paradoxically reduces protease binding affinity; this counterintuitive mechanism enhances metabolic stability.
C-terminal amidation further protects against carboxypeptidase activity; this modification eliminates the free carboxyl group that exopeptidases require for catalysis.
The combined PGP-C terminal and amidation strategies represent sophisticated peptide engineering; these modifications enable therapeutic utility that native sequences cannot achieve. Metabolic studies demonstrate intact Semax in circulation 6 to 8 hours post-administration; this persistence validates the structural protection approach.
Enkephalinase Inhibition: Molecular Mechanism and Clinical Relevance
Semax inhibits neutral endopeptidase (enkephalinase, neprilysin) with micromolar affinity; this inhibition elevates synaptic concentrations of endogenous opioid peptides. The enzyme cleaves the Gly-Phe bond of enkephalins; this cleavage terminates opioid peptide signaling at delta and mu receptors.
Semax binds the enzyme active site through competitive inhibition; this binding prevents substrate access without irreversible enzyme inactivation.
Total neural silence. That is the goal of a perfectly stabilized neuropeptide protocol.
Elevated enkephalin levels modulate mood, nociception, and stress responses; these effects complement the primary BDNF-mediated cognitive enhancement. The opioid peptide elevation remains within physiological ranges; this constraint prevents the respiratory depression and dependence associated with exogenous opioid agonists. Clinical observations describe anxiolytic effects distinct from the cognitive benefits; this dissociation suggests multiple independent mechanisms.
Enkephalinase inhibition occurs at lower concentrations than BDNF elevation; this differential potency enables mechanistic titration through dosing adjustments.
The dual mechanism provides broader therapeutic utility than either pathway alone; patients with anxiety-predominant symptoms may benefit disproportionately from the enkephalinase component. Chronic administration does not produce tolerance to either mechanism; this stability distinguishes Semax from traditional psychostimulants and opioid medications.
Semax: Technical Specifications
| Property | Value | Clinical Relevance |
|---|---|---|
| IUPAC Name | Met-Glu-His-Phe-Arg-Trp-Gly-Pro-Gly-Pro-NH2 | Heptapeptide with PGP C-terminal |
| Sequence | MEHFPGPPGP-NH2 | ACTH(4-10) extended with PGP |
| Weight | 813.97 g/mol | Optimal for BBB penetration |
| Formula | C37H51N11O10 | C-terminal amidated |
| CAS Number | 80714-61-0 | Standard reference identifier |
| Primary Targets | BDNF/trkB, Enkephalinase | Dual neurotrophic/opioid pathway |
| Bioavailability | 60-80% (intranasal) | Superior peptide absorption |
| Half-Life | 4-6 hours | Extended vs. native ACTH |
| Dosing Range | 300-600 mcg daily | Microgram-level peptide dosing |
Comparative Pharmacology: Semax versus Related Compounds
| Parameter | Semax | Adamax | Phenylpiracetam |
|---|---|---|---|
| Class | ACTH(4-7) analog | ACTH(4-10) adamantane | Phenylpyrrolidone |
| Mechanism | BDNF/enkephalinase | MC4R modulation | Dopamine/AMPA |
| Protection | PGP C-Terminal | Adamantane Cage | Phenyl Group |
| Duration | 12-24 hours | 8-12 hours | 4-6 hours |
| Stimulation | Minimal | Moderate | Significant |
The comparison reveals distinct mechanistic profiles; Semax targets enkephalinase and BDNF while Adamax focuses on melanocortin receptors.
Racetam-class compounds including phenylpiracetam operate through different pharmacological mechanisms; this diversity enables strategic selection based on individual requirements. Selank represents another Russian peptide with anxiolytic properties; this compound shares structural similarities with Semax but produces distinct clinical effects.
Selection between these compounds depends on therapeutic goals; Semax suits those prioritizing sustained cognitive enhancement with anxiolytic benefits. The PGP C-terminal modification distinguishes Semax metabolically; this distinction influences duration of action and dosing frequency. Clinical experience suggests individual response testing; some subjects respond optimally to Semax while others prefer Adamax or phenylpiracetam.
Clinical Applications and Stacking Considerations
Semax demonstrates particular utility for cognitive enhancement in demanding intellectual contexts; the sustained BDNF elevation supports complex problem-solving and memory consolidation. Cholinergic precursors complement the neurotrophic mechanisms; this combination addresses both synaptic substrate availability and trophic support.
Stacking with uridine monophosphate enhances phospholipid membrane synthesis; this enhancement provides structural substrate for plasticity-promoting effects.
Frankly, the data suggests that we are entering a new era of “Surgical Biohacking” where neuropeptides are the primary tool for neural resilience.
The compound shows promise for post-stroke cognitive rehabilitation; neuroprotective properties complement the cognitive enhancement mechanisms. N-acetylcysteine provides complementary antioxidant support; this combination addresses oxidative stress that impairs neuroplasticity. Long-term safety data from Russian clinical experience supports extended use protocols; however, Western regulatory approval remains pending.
Research Status and Pharmacovigilance
Semax research remains predominantly within Russian and Eastern European scientific literature; Western peer-reviewed publications remain limited though growing. The compound has not undergone FDA or EMA evaluation; this status restricts clinical availability outside research and compounded pharmacy contexts. Russian nootropic peptides consistently face regulatory barriers in Western markets; these barriers reflect historical publication patterns rather than safety concerns.
Mechanistic studies demonstrate consistent effects across multiple model systems; these findings support the theoretical basis for cognitive enhancement claims. Safety data from decades of Russian clinical use supports therapeutic index assessment; adverse event rates remain exceptionally low.
Future research priorities include controlled clinical trials in Western populations; such studies would establish efficacy and safety profiles for broader clinical acceptance.
Clinical Anecdotes & Human Biohacking Experience
Self-reported experiences from research chemical communities provide qualitative insight into Semax subjective effects; these unfiltered reports complement formal clinical literature. Individual responses demonstrate significant variation; baseline neurochemistry and dosing protocols influence outcomes substantially.
“bro semax is f***in legit. been on it 3 weeks now. my verbal fluency is NOTICEABLY better. like i can just… talk smoother? memory is sharper too. dosing 300mcg intranasal every morning. no sides at all. way cleaner than any racetam ive tried.”
“So I’ve been experimenting with Semax for about 2 months now. Started with 300mcg daily, bumped to 600mcg after week 2. The anxiety reduction was IMMEDIATE. Like day 1 I felt calmer. Cognitive benefits took maybe 5-7 days to really kick in. My focus during deep work sessions improved dramatically. Stacking with Alpha-GPC seems to amplify it. Only downside is the cost and sourcing reliability.”
“Idk what yall talking about Semax being subtle. For me it was STRONG. First dose felt like mild Adderall without the jitters. Clearheaded, motivated, social anxiety gone. Lasts all day too. 200mcg was my sweet spot. Higher doses made me irritable. Everyone’s different I guess.”
Clinical Synthesis
Semax represents a sophisticated example of peptide engineering; the PGP C-terminal modification provides metabolic stability that native ACTH fragments cannot achieve. Dual mechanisms of BDNF elevation and enkephalinase inhibition produce complementary cognitive and anxiolytic effects; this combination distinguishes Semax from single-mechanism compounds. The extensive Russian clinical experience supports therapeutic utility; however, Western practitioners lack familiarity with this evidence base.
Individual response testing remains fundamental; genetic variation in BDNF expression and enkephalinase activity influences clinical outcomes.
Stacking strategies should address complementary mechanisms; cholinergic support and phospholipid precursors provide structural substrate for the neuroplasticity-promoting effects. Quality sourcing presents practical challenges; the peptide requires refrigerated storage and careful handling to maintain potency.
The compound warrants consideration for cognitive enhancement protocols; the sustained BDNF elevation and anxiolytic properties address common clinical needs.
Comparison with adamantane-modified ACTH analogs reveals distinct mechanistic profiles; selection between these compounds depends on individual response and therapeutic goals. Further research should characterize optimal dosing and cycling protocols; current evidence supports flexible administration schedules.
Molecular Interactions with Neurotransmitter Systems
Semax demonstrates complex interactions with monoaminergic neurotransmission; these interactions extend beyond the primary BDNF and enkephalinase mechanisms. Dopaminergic tone in mesocortical pathways shows modulation; this modulation contributes to the motivational and reward-processing improvements observed clinically. Serotonergic signaling remains largely unaffected at therapeutic doses; this selectivity distinguishes Semax from serotonergic nootropics that produce distinct side effect profiles.
Glutamatergic neurotransmission shows subtle enhancement through BDNF-mediated mechanisms; this enhancement supports synaptic plasticity without producing excitotoxicity.
The compound does not directly interact with NMDA or AMPA receptors; indirect modulation through neurotrophic factors provides safer long-term profiles. GABAergic inhibition remains unchanged; this stability avoids the sedation or cognitive blunting associated with GABAergic compounds.
Acetylcholine synthesis and release show modest enhancement; this effect complements the primary mechanisms and supports attentional processes. The cholinergic modulation occurs through BDNF-mediated upregulation of choline acetyltransferase; this indirect mechanism avoids the receptor desensitization associated with direct cholinergic agonists. Stacking with choline precursors provides synergistic support for this pathway.
Patient Selection and Contraindications
Optimal candidates for Semax therapy include individuals with mild cognitive impairment; the BDNF elevation addresses the neurotrophic deficits common in this population.
Anxiety disorders with cognitive components respond particularly well; the dual anxiolytic and cognitive mechanisms address both symptom domains. Age-related cognitive decline represents another indication; the neuroprotective properties may slow progression of neurodegenerative processes.
Contraindications include active substance use disorders; the enkephalinase modulation could theoretically interact with opioid dependence pathways. Psychotic spectrum disorders represent relative contraindications; the neurotrophic effects require careful monitoring in these populations. Pregnancy and lactation remain absolute contraindications; insufficient safety data precludes risk assessment.
Drug interactions remain minimal; Semax does not significantly affect cytochrome P450 enzyme systems. Concurrent use with other peptides requires careful consideration; additive effects on neurotrophic pathways may produce unpredictable responses. Monitoring recommendations include baseline cognitive assessment and periodic efficacy evaluation; this surveillance ensures optimal therapeutic outcomes.
The clinical utility of Semax extends beyond simple cognitive enhancement; the compound addresses fundamental neurobiological processes that underlie neural resilience and plasticity. Long-term safety data from Russian clinical experience supports extended administration protocols; however, Western practitioners should approach with appropriate caution pending broader regulatory review.


0 Comments